Maturation of synaptic partners: functional phenotype and synaptic organization tuned in synchrony
- PMID: 20855433
- PMCID: PMC3008845
- DOI: 10.1113/jphysiol.2010.198564
Maturation of synaptic partners: functional phenotype and synaptic organization tuned in synchrony
Abstract
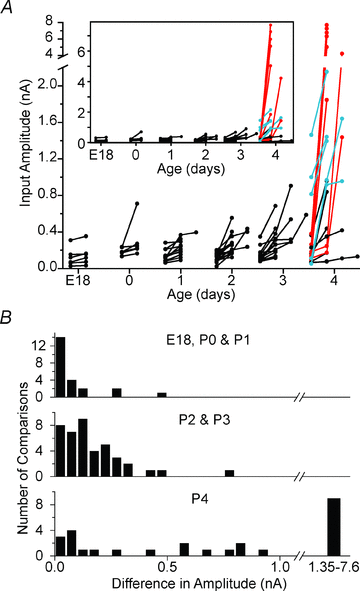
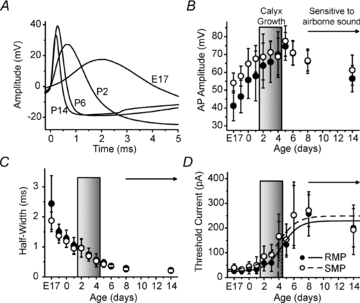
Maturation of principal neurons of the medial nucleus of the trapezoid body (MNTB) was assessed in the context of the developmental organization and activity of their presynaptic afferents, which grow rapidly to form calyces of Held and to establish mono-innervation between postnatal days (P)2 and 4. MNTB neurons and their inputs were studied from embryonic day (E)17, when the nucleus was first discernable, until P14 after the onset of hearing. Using a novel slice preparation containing portions of the cochlea, cochlear nucleus and MNTB, we determined that synaptic inputs form onto MNTB neurons at E17 and stimulation of the cochlear nucleus can evoke action potentials (APs) and Ca(2+) signals. We analysed converging inputs onto individual MNTB neurons and found that competition among inputs was resolved quickly, as a single large input, typically larger than 4 nA, emerged from P3-P4. During calyx growth but before hearing onset, MNTB cells acquired their mature, phasic firing property and quantitative real-time PCR confirmed a coincident increase in low threshold K(+) channel mRNA. These events occurred in concert with an increase in somatic surface area and a 7-fold increase in the current threshold (30 to >200 pA) required to evoke action potentials, as input resistance (R(in)) settled from embryonic values greater than 1 GΩ to approximately 200 MΩ. We postulate that the postsynaptic transition from hyperexcitability to decreased excitability during calyx growth could provide a mechanism to establish the mature 1:1 innervation by selecting the winning calyceal input based on synaptic strength. By comparing biophysical maturation of the postsynaptic cell to alterations in presynaptic organization, we propose that maturation of synaptic partners is coordinated by synaptic activity in a process that is likely to generalize to other neural systems.
Figures








Comment in
-
Early dating influences long-term synaptic partnerships.J Physiol. 2010 Nov 15;588(Pt 22):4339-40. doi: 10.1113/jphysiol.2010.200089. J Physiol. 2010. PMID: 21078599 Free PMC article. Review. No abstract available.
Similar articles
-
In vivo matching of postsynaptic excitability with spontaneous synaptic inputs during formation of the rat calyx of Held synapse.J Physiol. 2017 Jan 1;595(1):207-231. doi: 10.1113/JP272780. Epub 2016 Sep 15. J Physiol. 2017. PMID: 27426483 Free PMC article.
-
Deafferentation weakens excitatory synapses in the developing central auditory system.Eur J Neurosci. 1997 Nov;9(11):2340-7. doi: 10.1111/j.1460-9568.1997.tb01651.x. Eur J Neurosci. 1997. PMID: 9464928
-
Inhibitory projections from the ventral nucleus of the trapezoid body to the medial nucleus of the trapezoid body in the mouse.Front Neural Circuits. 2014 Jul 29;8:83. doi: 10.3389/fncir.2014.00083. eCollection 2014. Front Neural Circuits. 2014. PMID: 25120436 Free PMC article.
-
Formation and maturation of the calyx of Held.Hear Res. 2011 Jun;276(1-2):70-8. doi: 10.1016/j.heares.2010.11.004. Epub 2010 Nov 18. Hear Res. 2011. PMID: 21093567 Free PMC article. Review.
-
The ion channels and synapses responsible for the physiological diversity of mammalian lower brainstem auditory neurons.Hear Res. 2019 May;376:33-46. doi: 10.1016/j.heares.2018.12.011. Epub 2018 Dec 26. Hear Res. 2019. PMID: 30606624 Review.
Cited by
-
Embryonic assembly of auditory circuits: spiral ganglion and brainstem.J Physiol. 2012 May 15;590(10):2391-408. doi: 10.1113/jphysiol.2011.226886. Epub 2012 Feb 27. J Physiol. 2012. PMID: 22371481 Free PMC article.
-
BMP signaling specifies the development of a large and fast CNS synapse.Nat Neurosci. 2013 Jul;16(7):856-64. doi: 10.1038/nn.3414. Epub 2013 May 26. Nat Neurosci. 2013. PMID: 23708139
-
Temporal patterns of gene expression during calyx of held development.Dev Neurobiol. 2016 Feb;76(2):166-89. doi: 10.1002/dneu.22306. Epub 2015 Jul 8. Dev Neurobiol. 2016. PMID: 26014473 Free PMC article.
-
Physiological Perspectives on Molecular Mechanisms and Regulation of Vesicular Glutamate Transport: Lessons From Calyx of Held Synapses.Front Cell Neurosci. 2022 Jan 13;15:811892. doi: 10.3389/fncel.2021.811892. eCollection 2021. Front Cell Neurosci. 2022. PMID: 35095427 Free PMC article. Review.
-
Presynaptic loss of dynamin-related protein 1 impairs synaptic vesicle release and recycling at the mouse calyx of Held.J Physiol. 2018 Dec;596(24):6263-6287. doi: 10.1113/JP276424. Epub 2018 Nov 10. J Physiol. 2018. PMID: 30285293 Free PMC article.
References
-
- Awatramani GB, Turecek R, Trussell LO. Staggered development of GABAergic and glycinergic transmission in the MNTB. J Neurophysiol. 2005;93:819–828. - PubMed
-
- Bewick GS, Reid B, Jawaid S, Hatcher T, Shanley L. Postnatal emergence of mature release properties in terminals of rat fast- and slow-twitch muscles. Eur J Neurosci. 2004;19:2967–2976. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

