Orally administered particulate beta-glucan modulates tumor-capturing dendritic cells and improves antitumor T-cell responses in cancer
- PMID: 20855461
- PMCID: PMC2970627
- DOI: 10.1158/1078-0432.CCR-10-0820
Orally administered particulate beta-glucan modulates tumor-capturing dendritic cells and improves antitumor T-cell responses in cancer
Abstract
Purpose: The beneficial properties of β-glucans have been recognized for centuries. Their proposed mechanisms of action in cancer therapy occur via stimulation of macrophages and priming of innate neutrophil complement receptor 3 for eliciting complement receptor 3-dependent cellular cytotoxicity of iC3b-opsonized tumor cells. The current study is to investigate whether β-glucan therapy has any effect on antitumor adaptive T-cell responses.
Experimental design: We first examined the trafficking of orally administered particulate yeast-derived β-glucan and its interaction with dendritic cells (DC) that captured tumor materials. Antigen-specific T cells were adoptively transferred into recipient mice to determine whether oral β-glucan therapy induces augmented T-cell responses. Lewis lung carcinoma and RAM-S lymphoma models were used to test oral β-glucan therapeutic effect. Further mechanistic studies including tumor-infiltrating T cells and cytokine profiles within the tumor milieu were determined.
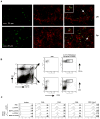
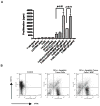
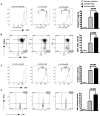
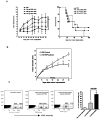
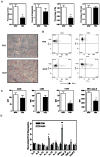
Results: Orally administered particulate β-glucan trafficked into spleen and lymph nodes and activated DCs that captured dying tumor cells in vivo, leading to the expansion and activation of antigen-specific CD4 and CD8 T cells. In addition, IFN-γ production of tumor-infiltrating T cells and CTL responses were significantly enhanced on β-glucan treatment, which ultimately resulted in significantly reduced tumor burden. Moreover, β-glucan-treated tumors had significantly more DC infiltration with the activated phenotype and significant levels of Th1-biased cytokines within the tumor microenvironment.
Conclusions: These data highlight the ability of yeast-derived β-glucan to bridge innate and adaptive antitumor immunity and suggest that it can be used as an adjuvant for tumor immunotherapy.
©2010 AACR.
Figures
Similar articles
-
Differential pathways regulating innate and adaptive antitumor immune responses by particulate and soluble yeast-derived β-glucans.Blood. 2011 Jun 23;117(25):6825-36. doi: 10.1182/blood-2011-02-339812. Epub 2011 Apr 29. Blood. 2011. PMID: 21531981 Free PMC article.
-
β-glucan from Aureobasidium pullulans augments the anti-tumor immune responses through activated tumor-associated dendritic cells.Int Immunopharmacol. 2021 Dec;101(Pt A):108265. doi: 10.1016/j.intimp.2021.108265. Epub 2021 Oct 29. Int Immunopharmacol. 2021. PMID: 34715491
-
β-Glucan enhances antitumor immune responses by regulating differentiation and function of monocytic myeloid-derived suppressor cells.Eur J Immunol. 2013 May;43(5):1220-30. doi: 10.1002/eji.201242841. Epub 2013 Mar 25. Eur J Immunol. 2013. PMID: 23424024
-
The effects of β-glucans on dendritic cells and implications for cancer therapy.Anticancer Agents Med Chem. 2013 Jun;13(5):689-98. doi: 10.2174/1871520611313050003. Anticancer Agents Med Chem. 2013. PMID: 23092290 Review.
-
Consumption of β-glucans to spice up T cell treatment of tumors: a review.Expert Opin Biol Ther. 2018 Oct;18(10):1023-1040. doi: 10.1080/14712598.2018.1523392. Expert Opin Biol Ther. 2018. PMID: 30221551 Review.
Cited by
-
Trained immunity inducers in cancer immunotherapy.Front Immunol. 2024 Jul 16;15:1427443. doi: 10.3389/fimmu.2024.1427443. eCollection 2024. Front Immunol. 2024. PMID: 39081326 Free PMC article. Review.
-
Heat-killed Mycobacterium tuberculosis induces trained immunity in vitro and in vivo administered systemically or intranasally.iScience. 2024 Jan 11;27(2):108869. doi: 10.1016/j.isci.2024.108869. eCollection 2024 Feb 16. iScience. 2024. PMID: 38318361 Free PMC article.
-
Functional immune boosters; the herb or its dead microbiome? Antigenic TLR4 agonist MAMPs found in 65 medicinal roots and algae's.J Funct Foods. 2023 Aug;107:105687. doi: 10.1016/j.jff.2023.105687. Epub 2023 Jul 29. J Funct Foods. 2023. PMID: 37654434 Free PMC article.
-
Postbiotics-A Step Beyond Pre- and Probiotics.Nutrients. 2020 Jul 23;12(8):2189. doi: 10.3390/nu12082189. Nutrients. 2020. PMID: 32717965 Free PMC article. Review.
-
A promising Staphylococcus aureus vaccine adjuvant candidate to overcome Staphylococcal infection based on innate immunity.Clin Exp Vaccine Res. 2025 Jul;14(3):189-202. doi: 10.7774/cevr.2025.14.e27. Epub 2025 Jun 13. Clin Exp Vaccine Res. 2025. PMID: 40741061 Free PMC article. Review.
References
-
- Diller IC, Mankowski ZT, Fisher ME. The effect of yeast polysaccharides on mouse tumors. Cancer Res. 1963;23:201–8. - PubMed
-
- Di Luzio NR, Williams DL, McNamee RB, Edwards BF, Kitahama A. Comparative tumor-inhibitory and anti-bacterial activity of soluble and particulate glucan. Int J Cancer. 1979;24:773–9. - PubMed
-
- Hunter JT, Meltzer MS, Ribi E, Fidler IJ, Hanna MG, Jr, Zbar B, et al. Glucan: attempts to demonstrate therapeutic activity against five syngeneic tumors in guinea pigs and mice. J Natl Cancer Inst. 1978;60:419–24. - PubMed
-
- Lavigne LM, O’Brien XM, Kim M, Janowski JW, Albina JE, Reichner JS. Integrin engagement mediates the human polymorphonuclear leukocyte response to a fungal pathogen-associated molecular pattern. J Immunol. 2007;178:7276–82. - PubMed
-
- Ensley HE, Tobias B, Pretus HA, McNamee RB, Jones EL, Browder IW, et al. NMR spectral analysis of a water-insoluble (1-->3)-beta-D-glucan isolated from Saccharomyces cerevisiae. Carbohydr Res. 1994;258:307–11. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

