Znu is the predominant zinc importer in Yersinia pestis during in vitro growth but is not essential for virulence
- PMID: 20855510
- PMCID: PMC2981304
- DOI: 10.1128/IAI.00732-10
Znu is the predominant zinc importer in Yersinia pestis during in vitro growth but is not essential for virulence
Abstract
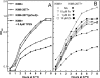
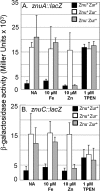
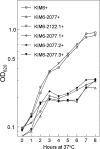
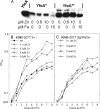
Little is known about Zn homeostasis in Yersinia pestis, the plague bacillus. The Znu ABC transporter is essential for zinc (Zn) uptake and virulence in a number of bacterial pathogens. Bioinformatics analysis identified ZnuABC as the only apparent high-affinity Zn uptake system in Y. pestis. Mutation of znuACB caused a growth defect in Chelex-100-treated PMH2 growth medium, which was alleviated by supplementation with submicromolar concentrations of Zn. Use of transcriptional reporters confirmed that Zur mediated Zn-dependent repression and that it can repress gene expression in response to Zn even in the absence of Znu. Virulence testing in mouse models of bubonic and pneumonic plague found only a modest increase in survival in low-dose infections by the znuACB mutant. Previous studies of cluster 9 (C9) transporters suggested that Yfe, a well-characterized C9 importer for manganese (Mn) and iron in Y. pestis, might function as a second, high-affinity Zn uptake system. Isothermal titration calorimetry revealed that YfeA, the solute-binding protein component of Yfe, binds Mn and Zn with comparably high affinities (dissociation constants of 17.8 ± 4.4 nM and 6.6 ± 1.2 nM, respectively), although the complete Yfe transporter could not compensate for the loss of Znu in in vitro growth studies. Unexpectedly, overexpression of Yfe interfered with the znu mutant's ability to grow in low concentrations of Zn, while excess Zn interfered with the ability of Yfe to import iron at low concentrations; these results suggest that YfeA can bind Zn in the bacterial cell but that Yfe is incompetent for transport of the metal. In addition to Yfe, we have now eliminated MntH, FetMP, Efe, Feo, a substrate-binding protein, and a putative nickel transporter as the unidentified, secondary Zn transporter in Y. pestis. Unlike other bacterial pathogens, Y. pestis does not require Znu for high-level infectivity and virulence; instead, it appears to possess a novel class of transporter, which can satisfy the bacterium's Zn requirements under in vivo metal-limiting conditions. Our studies also underscore the need for bacterial cells to balance binding and transporter specificities within the periplasm in order to maintain transition metal homeostasis.
Figures


Similar articles
-
Manganese transporters Yfe and MntH are Fur-regulated and important for the virulence of Yersinia pestis.Microbiology (Reading). 2012 Mar;158(Pt 3):804-815. doi: 10.1099/mic.0.053710-0. Epub 2012 Jan 5. Microbiology (Reading). 2012. PMID: 22222497 Free PMC article.
-
Zinc transporters YbtX and ZnuABC are required for the virulence of Yersinia pestis in bubonic and pneumonic plague in mice.Metallomics. 2017 Jun 21;9(6):757-772. doi: 10.1039/c7mt00126f. Metallomics. 2017. PMID: 28540946 Free PMC article.
-
The Yfe and Feo transporters are involved in microaerobic growth and virulence of Yersinia pestis in bubonic plague.Infect Immun. 2012 Nov;80(11):3880-91. doi: 10.1128/IAI.00086-12. Epub 2012 Aug 27. Infect Immun. 2012. PMID: 22927049 Free PMC article.
-
The role of transition metal transporters for iron, zinc, manganese, and copper in the pathogenesis of Yersinia pestis.Metallomics. 2015 Jun;7(6):965-78. doi: 10.1039/c4mt00332b. Metallomics. 2015. PMID: 25891079 Free PMC article. Review.
-
Genetic Regulation of Yersinia pestis.Adv Exp Med Biol. 2016;918:223-256. doi: 10.1007/978-94-024-0890-4_8. Adv Exp Med Biol. 2016. PMID: 27722865 Review.
Cited by
-
Identification, Functional Characterization, and Regulon Prediction of the Zinc Uptake Regulator (zur) of Bacillus anthracis - An Insight Into the Zinc Homeostasis of the Pathogen.Front Microbiol. 2019 Jan 11;9:3314. doi: 10.3389/fmicb.2018.03314. eCollection 2018. Front Microbiol. 2019. PMID: 30687290 Free PMC article.
-
Acclimation to Nutritional Immunity and Metal Intoxication Requires Zinc, Manganese, and Copper Homeostasis in the Pathogenic Neisseriae.Front Cell Infect Microbiol. 2022 Jun 30;12:909888. doi: 10.3389/fcimb.2022.909888. eCollection 2022. Front Cell Infect Microbiol. 2022. PMID: 35846739 Free PMC article. Review.
-
Manganese transporters Yfe and MntH are Fur-regulated and important for the virulence of Yersinia pestis.Microbiology (Reading). 2012 Mar;158(Pt 3):804-815. doi: 10.1099/mic.0.053710-0. Epub 2012 Jan 5. Microbiology (Reading). 2012. PMID: 22222497 Free PMC article.
-
Coregulation of host-adapted metabolism and virulence by pathogenic yersiniae.Front Cell Infect Microbiol. 2014 Oct 20;4:146. doi: 10.3389/fcimb.2014.00146. eCollection 2014. Front Cell Infect Microbiol. 2014. PMID: 25368845 Free PMC article. Review.
-
Zinc limitation in Klebsiella pneumoniae profiled by quantitative proteomics influences transcriptional regulation and cation transporter-associated capsule production.BMC Microbiol. 2021 Feb 10;21(1):43. doi: 10.1186/s12866-021-02091-8. BMC Microbiol. 2021. PMID: 33568055 Free PMC article.
References
-
- Ammendola, S., P. Pasquali, C. Pistoia, P. Petrucci, P. Petrarca, G. Rotilio, and A. Battistoni. 2007. High-affinity Zn2+ uptake system ZnuABC is required for bacterial zinc homeostasis in intracellular environments and contributes to the virulence of Salmonella enterica. Infect. Immun. 75:5867-5876. - PMC - PubMed
-
- Anderson, E. S., J. T. Paulley, J. M. Gaines, M. W. Valderas, D. W. Martin, E. Menscher, T. D. Brown, C. S. Burns, and R. M. Roop II. 2009. The manganese transporter MntH is a critical virulence determinant for Brucella abortus 2308 in experimentally infected mice. Infect. Immun. 77:3466-3474. - PMC - PubMed
-
- Andreini, C., L. Banci, I. Bertini, and A. Rosato. 2006. Zinc through the three domains of life. J. Proteome Res. 5:3173-3178. - PubMed
-
- Arnold, K., L. Bordoli, J. Kopp, and T. Schwede. 2006. The SWISS-MODEL workspace: a web-based environment for protein structure homology modeling. Bioinformatics 22:195-201. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

