Cutting edge: TCR ligation triggers digital activation of NF-kappaB
- PMID: 20855880
- PMCID: PMC2950878
- DOI: 10.4049/jimmunol.1001051
Cutting edge: TCR ligation triggers digital activation of NF-kappaB
Abstract
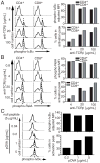
TCR-mediated activation of the transcription factor NF-κB is required for T cell proliferation, survival, and effector differentiation. Although this pathway is the subject of intense study, it is not known whether TCR signaling to NF-κB is digital (switch-like) or analog in nature. Through analysis of the phosphorylation and degradation of IκBα and the nuclear translocation and phosphorylation of the NF-κB subunit RelA, we show that TCR-directed NF-κB activation is digital. Furthermore, digitization occurs well upstream of the IκB kinase complex, as protein kinase C translocation to the immunologic synapse and activation-associated aggregation of Bcl10 and Malt1 also demonstrate both digital behavior and high correlation with RelA nuclear translocation. Thus, similar to the TCR-to-MAPK signaling cascade, analog Ag inputs are converted to digital activation outputs to NF-κB at an early step downstream of TCR ligation.
Figures


References
-
- Tian T, Harding A, Inder K, Plowman S, Parton RG, Hancock JF. Plasma membrane nanoswitches generate high-fidelity Ras signal transduction. Nat Cell Biol. 2007;9:905–914. - PubMed
-
- Stefanova I, Hemmer B, Vergelli M, Martin R, Biddison WE, Germain RN. TCR ligand discrimination is enforced by competing ERK positive and SHP-1 negative feedback pathways. Nat Immunol. 2003;4:248–254. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

