The use of PEGylated liposomes in the development of drug delivery applications for the treatment of hemophilia
- PMID: 20856833
- PMCID: PMC2939703
- DOI: 10.2147/ijn.s8603
The use of PEGylated liposomes in the development of drug delivery applications for the treatment of hemophilia
Abstract
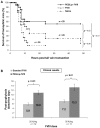
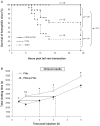
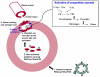
Hemophilia A is a rare X-linked bleeding disorder caused by lack or dysfunction of coagulation factor VIII (FVIII). Hemophilia A is treated with replacement therapy, but frequent injections of the missing FVIII often lead to the formation of inhibitory antibodies. Patients who develop high levels of inhibitors must be treated with bypassing agents such as activated FVII (FVIIa). Both FVIII and FVIIa have short half-lives and require multiple injections. Long-acting forms of these proteins would therefore reduce the frequency of injections, improve patient compliance and reduce complications. In this article we present a new platform technology that produces long-acting forms of FVIII and FVIIa and improves the efficacy of hemophilia treatment. This technology is based on the binding of proteins/peptides to the outer surface of PEGylated liposomes (PEGLip). Binding is dependent on an amino acid consensus sequence within the proteins and is highly specific. At the same time, binding is non-covalent and does not require any modification of the therapeutic agent or its production process. Association of proteins with PEGLip results in substantial enhancements in their pharmacodynamic properties following administration. These improvements seem to arise from the association of formulated proteins with platelets prior to induction of coagulation.
Keywords: PEGylated liposomes; factor VIII; factor VIIa; pharmacodynamics; therapeutic proteins.
Figures

Similar articles
-
Safety, pharmacokinetics and efficacy of factor VIIa formulated with PEGylated liposomes in haemophilia A patients with inhibitors to factor VIII--an open label, exploratory, cross-over, phase I/II study.Haemophilia. 2010 Nov;16(6):910-8. doi: 10.1111/j.1365-2516.2010.02273.x. Haemophilia. 2010. PMID: 20491957 Clinical Trial.
-
Enhancement of the efficacy of therapeutic proteins by formulation with PEGylated liposomes; a case of FVIII, FVIIa and G-CSF.Expert Opin Drug Deliv. 2010 Feb;7(2):187-201. doi: 10.1517/17425240903463846. Expert Opin Drug Deliv. 2010. PMID: 20095942 Review.
-
Factor VIII efficient and specific non-covalent binding to PEGylated liposomes enables prolongation of its circulation time and haemostatic efficacy.Thromb Haemost. 2005 Jun;93(6):1061-8. doi: 10.1160/TH04-08-0485. Thromb Haemost. 2005. PMID: 15968389
-
Enhancement of factor VIIa haemostatic efficacy by formulation with PEGylated liposomes.Haemophilia. 2008 May;14(3):476-83. doi: 10.1111/j.1365-2516.2008.01741.x. Epub 2008 Apr 3. Haemophilia. 2008. PMID: 18393980
-
Gene-based continuous expression of FVIIa for the treatment of hemophilia.Front Biosci (Schol Ed). 2012 Jan 1;4(1):287-99. doi: 10.2741/s269. Front Biosci (Schol Ed). 2012. PMID: 22202061 Review.
Cited by
-
Curcumin Encapsulated PEGylated Nanoliposomes: A Potential Anti-Infective Therapeutic Agent.Indian J Microbiol. 2019 Sep;59(3):336-343. doi: 10.1007/s12088-019-00811-3. Epub 2019 Jun 7. Indian J Microbiol. 2019. PMID: 31388211 Free PMC article.
-
Study of toxicity effect of pegylated nanoliposomal artemisinin on breast cancer cell line.Indian J Clin Biochem. 2013 Oct;28(4):410-2. doi: 10.1007/s12291-013-0306-3. Epub 2013 Feb 10. Indian J Clin Biochem. 2013. PMID: 24426245 Free PMC article.
-
Mind the gap: a survey of how cancer drug carriers are susceptible to the gap between research and practice.J Control Release. 2013 Dec 28;172(3):1045-64. doi: 10.1016/j.jconrel.2013.09.026. Epub 2013 Oct 2. J Control Release. 2013. PMID: 24096014 Free PMC article. Review.
-
Therapeutic and routine prophylactic properties of rFactor VIII Fc (efraloctocog alfa, Eloctate®) in hemophilia A.J Blood Med. 2016 Sep 12;7:187-198. doi: 10.2147/JBM.S80814. eCollection 2016. J Blood Med. 2016. PMID: 27695377 Free PMC article. Review.
-
In vitro evaluation of the efficacy of liposomal and pegylated liposomal hydroxyurea.Indian J Clin Biochem. 2014 Jan;29(1):84-8. doi: 10.1007/s12291-013-0315-2. Epub 2013 Mar 26. Indian J Clin Biochem. 2014. PMID: 24478555 Free PMC article.
References
-
- Dahlback B. Blood coagulation and its regulation by anticoagulant pathways: genetic pathogenesis of bleeding and thrombotic diseases. J Intern Med. 2005;257:209–223. - PubMed
-
- Hedner U, Ginsburg D, Lusher JM, High KA. Congenital Hemorrhagic Disorders: New Insights into the Pathophysiology and Treatment of Hemophilia. Hematology Am Soc Hematol Educ Program. 2000:241–265. - PubMed
-
- Soucie JM, Evatt B, Jackson D. Occurrence of hemophilia in the United States. The Hemophilia Surveillance System Project Investigators. Am J Hematol. 1998;59:288–294. - PubMed
-
- Kaufman RJ, Anthonorakis SE, Fay PJ. Hemostasis and Thrombosis: Basic Principles and Clinical Practice. 4th ed. Philadelphia: Lippincott Williams and Wilkins; 2001.
-
- Roosendaal G, Mauser-Bunschoten EP, De Kleijn P, et al. Synovium in haemophilic arthropathy. Haemophilia. 1998;4:502–505. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

