PeptideMine--a webserver for the design of peptides for protein-peptide binding studies derived from protein-protein interactomes
- PMID: 20858292
- PMCID: PMC2955050
- DOI: 10.1186/1471-2105-11-473
PeptideMine--a webserver for the design of peptides for protein-peptide binding studies derived from protein-protein interactomes
Abstract
Background: Signal transduction events often involve transient, yet specific, interactions between structurally conserved protein domains and polypeptide sequences in target proteins. The identification and validation of these associating domains is crucial to understand signal transduction pathways that modulate different cellular or developmental processes. Bioinformatics strategies to extract and integrate information from diverse sources have been shown to facilitate the experimental design to understand complex biological events. These methods, primarily based on information from high-throughput experiments, have also led to the identification of new connections thus providing hypothetical models for cellular events. Such models, in turn, provide a framework for directing experimental efforts for validating the predicted molecular rationale for complex cellular processes. In this context, it is envisaged that the rational design of peptides for protein-peptide binding studies could substantially facilitate the experimental strategies to evaluate a predicted interaction. This rational design procedure involves the integration of protein-protein interaction data, gene ontology, physico-chemical calculations, domain-domain interaction data and information on functional sites or critical residues.
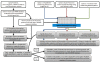
Results: Here we describe an integrated approach called "PeptideMine" for the identification of peptides based on specific functional patterns present in the sequence of an interacting protein. This approach based on sequence searches in the interacting sequence space has been developed into a webserver, which can be used for the identification and analysis of peptides, peptide homologues or functional patterns from the interacting sequence space of a protein. To further facilitate experimental validation, the PeptideMine webserver also provides a list of physico-chemical parameters corresponding to the peptide to determine the feasibility of using the peptide for in vitro biochemical or biophysical studies.
Conclusions: The strategy described here involves the integration of data and tools to identify potential interacting partners for a protein and design criteria for peptides based on desired biochemical properties. Alongside the search for interacting protein sequences using three different search programs, the server also provides the biochemical characteristics of candidate peptides to prune peptide sequences based on features that are most suited for a given experiment. The PeptideMine server is available at the URL: http://caps.ncbs.res.in/peptidemine.
Figures




References
-
- Shah PK, Tripathi LP, Jensen LJ, Gahnim M, Mason C, Furlong EE, Rodrigues V, White KP, Bork P, Sowdhamini R. Enhanced function annotations for Drosophila serine proteases: a case study for systematic annotation of multi-member gene families. Gene. 2008;407:199–215. doi: 10.1016/j.gene.2007.10.012. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

