The crystal structure of non-modified and bipyridine-modified PNA duplexes
- PMID: 20859960
- PMCID: PMC3194003
- DOI: 10.1002/chem.201000392
The crystal structure of non-modified and bipyridine-modified PNA duplexes
Erratum in
- Chemistry. 2011 Oct 24;17(44):12227
Abstract
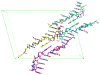
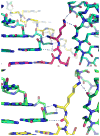
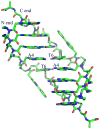
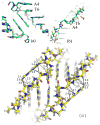
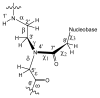
Peptide nucleic acid (PNA) is a synthetic analogue of DNA that commonly has an N-aminoethyl glycine backbone. The crystal structures of two PNA duplexes, one containing eight standard nucleobase pairs (GGCATGCC)(2), and the other containing the same nucleobase pairs and a central pair of bipyridine ligands, have been solved with a resolution of 1.22 and 1.10 Å, respectively. The non-modified PNA duplex adopts a P-type helical structure similar to that of previously characterized PNAs. The atomic-level resolution of the structures allowed us to observe for the first time specific modes of interaction between the terminal lysines of the PNA and the backbone and the nucleobases situated in the vicinity of the lysines, which are considered an important factor in the induction of a preferred handedness in PNA duplexes. Our results support the notion that whereas PNA typically adopts a P-type helical structure, its flexibility is relatively high. For example, the base-pair rise in the bipyridine-containing PNA is the largest measured to date in a PNA homoduplex. The two bipyridines bulge out of the duplex and are aligned parallel to the major groove of the PNA. In addition, two bipyridines from adjacent PNA duplexes form a π-stacked pair that relates the duplexes within the crystal. The bulging out of the bipyridines causes bending of the PNA duplex, which is in contrast to the structure previously reported for biphenyl-modified DNA duplexes in solution, where the biphenyls are π stacked with adjacent nucleobase pairs and adopt an intrahelical geometry. This difference shows that relatively small perturbations can significantly impact the relative position of nucleobase analogues in nucleic acid duplexes.
Figures
Similar articles
-
Metal incorporation in modified PNA duplexes.J Am Chem Soc. 2003 May 28;125(21):6354-5. doi: 10.1021/ja029714v. J Am Chem Soc. 2003. PMID: 12785760
-
Metal binding to bipyridine-modified PNA.Inorg Chem. 2006 Nov 27;45(24):9798-811. doi: 10.1021/ic0609610. Inorg Chem. 2006. PMID: 17112277
-
Coordination-driven inversion of handedness in ligand-modified PNA.Inorg Chem. 2011 Dec 5;50(23):11929-37. doi: 10.1021/ic200855p. Epub 2011 Nov 7. Inorg Chem. 2011. PMID: 22059624
-
Peptide-nucleic acids (PNAs): a tool for the development of gene expression modifiers.Curr Pharm Des. 2001 Nov;7(17):1839-62. doi: 10.2174/1381612013397087. Curr Pharm Des. 2001. PMID: 11562312 Review.
-
Helix control in polymers: case of peptide nucleic acids (PNAs).Artif DNA PNA XNA. 2012 Apr-Jun;3(2):31-44. doi: 10.4161/adna.20572. Epub 2012 Apr 1. Artif DNA PNA XNA. 2012. PMID: 22772039 Free PMC article. Review.
Cited by
-
The structural diversity of artificial genetic polymers.Nucleic Acids Res. 2016 Feb 18;44(3):1007-21. doi: 10.1093/nar/gkv1472. Epub 2015 Dec 15. Nucleic Acids Res. 2016. PMID: 26673703 Free PMC article. Review.
-
Antibacterial Peptide Nucleic Acids-Facts and Perspectives.Molecules. 2020 Jan 28;25(3):559. doi: 10.3390/molecules25030559. Molecules. 2020. PMID: 32012929 Free PMC article. Review.
-
Structural insight into DNA-assembled oligochromophores: crystallographic analysis of pyrene- and phenanthrene-modified DNA in complex with BpuJI endonuclease.Nucleic Acids Res. 2016 Sep 6;44(15):7079-89. doi: 10.1093/nar/gkw644. Epub 2016 Jul 15. Nucleic Acids Res. 2016. PMID: 27422870 Free PMC article.
-
The first crystal structures of RNA-PNA duplexes and a PNA-PNA duplex containing mismatches--toward anti-sense therapy against TREDs.Nucleic Acids Res. 2016 Feb 29;44(4):1937-43. doi: 10.1093/nar/gkv1513. Epub 2015 Dec 29. Nucleic Acids Res. 2016. PMID: 26717983 Free PMC article.
-
The proto-Nucleic Acid Builder: a software tool for constructing nucleic acid analogs.Nucleic Acids Res. 2021 Jan 11;49(1):79-89. doi: 10.1093/nar/gkaa1159. Nucleic Acids Res. 2021. PMID: 33300028 Free PMC article.
References
-
- Nielsen PE, Egholm M. Current Issues in Molecular Biology. 1999;1:89. - PubMed
-
- Nielsen PE, Egholm M, Berg RH, Buchardt O. Science. 1991;254:1497. - PubMed
-
- Achim C, Armitage BA, Ly DH, Schneider JW. Wiley Encyclopedia of Chemical Biology. John Wiley & Sons; 2008.
-
- Rasmussen H, Kastrup JS, Nielsen JN, Nielsen JM, Nielsen PE. Nat Struct Biol. 1997;4:98. - PubMed
-
- Rasmussen H, Liljefors T, Petersson B, Nielsen PE, Kastrup JS. J Biomol Struct Dyn. 2004;21:495. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

