Jen1p: a high affinity selenite transporter in yeast
- PMID: 20861301
- PMCID: PMC2982120
- DOI: 10.1091/mbc.E10-06-0513
Jen1p: a high affinity selenite transporter in yeast
Abstract
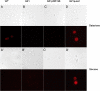
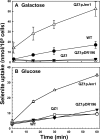
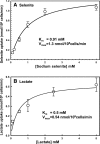
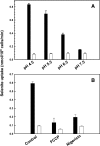
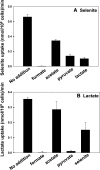
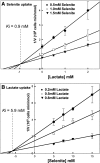
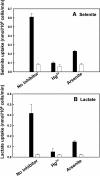
Selenium is a micronutrient in most eukaryotes, including humans, which is well known for having an extremely thin border between beneficial and toxic concentrations. Soluble tetravalent selenite is the predominant environmental form and also the form that is applied in the treatment of human diseases. To acquire this nutrient from low environmental concentrations as well as to avoid toxicity, a well-controlled transport system is required. Here we report that Jen1p, a proton-coupled monocarboxylate transporter in S. cerevisiae, catalyzes high-affinity uptake of selenite. Disruption of JEN1 resulted in selenite resistance, and overexpression resulted in selenite hypersensitivity. Transport assay showed that overexpression of Jen1p enables selenite accumulation in yeast compared with a JEN1 knock out strain, indicating the Jen1p transporter facilitates selenite accumulation inside cells. Selenite uptake by Jen1p had a Km of 0.91 mM, which is comparable to the Km for lactate. Jen1p transported selenite in a proton-dependent manner which resembles the transport mechanism for lactate. In addition, selenite and lactate can inhibit the transport of each other competitively. Therefore, we postulate selenite is a molecular mimic of monocarboxylates which allows selenite to be transported by Jen1p.
Figures


Similar articles
-
Functional expression of the lactate permease Jen1p of Saccharomyces cerevisiae in Pichia pastoris.Biochem J. 2003 Dec 15;376(Pt 3):781-7. doi: 10.1042/BJ20031180. Biochem J. 2003. PMID: 12962538 Free PMC article.
-
The conserved sequence NXX[S/T]HX[S/T]QDXXXT of the lactate/pyruvate:H(+) symporter subfamily defines the function of the substrate translocation pathway.Mol Membr Biol. 2007 Sep-Dec;24(5-6):464-74. doi: 10.1080/09687680701342669. Mol Membr Biol. 2007. PMID: 17710650
-
Improvement of ethanol production from D-lactic acid by constitutive expression of lactate transporter Jen1p in Saccharomyces cerevisiae.Biosci Biotechnol Biochem. 2013;77(5):1114-6. doi: 10.1271/bbb.120985. Epub 2013 May 7. Biosci Biotechnol Biochem. 2013. PMID: 23649240
-
Carboxylic Acids Plasma Membrane Transporters in Saccharomyces cerevisiae.Adv Exp Med Biol. 2016;892:229-251. doi: 10.1007/978-3-319-25304-6_9. Adv Exp Med Biol. 2016. PMID: 26721276 Review.
-
Carbon source dependent promoters in yeasts.Microb Cell Fact. 2014 Jan 9;13:5. doi: 10.1186/1475-2859-13-5. Microb Cell Fact. 2014. PMID: 24401081 Free PMC article. Review.
Cited by
-
Characterization of a Novel Porin-Like Protein, ExtI, from Geobacter sulfurreducens and Its Implication in the Reduction of Selenite and Tellurite.Int J Mol Sci. 2018 Mar 11;19(3):809. doi: 10.3390/ijms19030809. Int J Mol Sci. 2018. PMID: 29534491 Free PMC article.
-
ATP synthesis in the energy metabolism pathway: a new perspective for manipulating CdSe quantum dots biosynthesized in Saccharomyces cerevisiae.Int J Nanomedicine. 2017 May 19;12:3865-3879. doi: 10.2147/IJN.S132719. eCollection 2017. Int J Nanomedicine. 2017. PMID: 28579774 Free PMC article.
-
Mechanisms underlying lactic acid tolerance and its influence on lactic acid production in Saccharomyces cerevisiae.Microb Cell. 2021 Apr 14;8(6):111-130. doi: 10.15698/mic2021.06.751. Microb Cell. 2021. PMID: 34055965 Free PMC article. Review.
-
Engineering Flocculation for Improved Tolerance and Production of d-Lactic Acid in Pichia pastoris.J Fungi (Basel). 2023 Mar 27;9(4):409. doi: 10.3390/jof9040409. J Fungi (Basel). 2023. PMID: 37108864 Free PMC article.
-
High-Throughput Profiling of Candida auris Isolates Reveals Clade-Specific Metabolic Differences.Microbiol Spectr. 2023 Jun 15;11(3):e0049823. doi: 10.1128/spectrum.00498-23. Epub 2023 Apr 25. Microbiol Spectr. 2023. PMID: 37097196 Free PMC article. Review.
References
-
- Akita O., Nishimori C., Shimamoto T., Fujii T., Iefuji H. Transport of pyruvate in Saccharomyces cerevisiae and cloning of the gene encoded pyruvate permease. Biosci Biotechnol Biochem. 2000;64:980–984. - PubMed
-
- Blessing H., Kraus S., Heindl P., Bal W., Hartwig A. Interaction of selenium compounds with zinc finger proteins involved in DNA repair. Eur. J. Biochem. 2004;271:3190–3199. - PubMed
-
- Bojunga N., Entian K. D. Cat8p, the activator of gluconeogenic genes in Saccharomyces cerevisiae, regulates carbon source-dependent expression of NADP-dependent cytosolic isocitrate dehydrogenase (Idp2p) and lactate permease (Jen1p) Mol. Gen. Genet. 1999;262:869–875. - PubMed
-
- Brown T. A., Shrift A. Assimilation of selenate and selenite by Salmonella typhimurium. Can. J. Microbiol. 1980;26:671–675. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

