The behavioral neuroscience of anuran social signal processing
- PMID: 20863685
- PMCID: PMC3010340
- DOI: 10.1016/j.conb.2010.08.021
The behavioral neuroscience of anuran social signal processing
Abstract
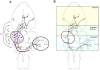
Acoustic communication is the major component of social behavior in anuran amphibians (frogs and toads) and has served as a neuroethological model for the nervous system's processing of social signals related to mate choice decisions. The male's advertisement or mating call is its most conspicuous social signal, and the nervous system's analysis of the call is a progressive process. As processing proceeds through neural systems, response properties become more specific to the signal and, in addition, neural activity gradually shifts from representing sensory (auditory periphery and brainstem) to sensorimotor (diencephalon) to motor (forebrain) components of a behavioral response. A comparative analysis of many anuran species shows that the first stage in biasing responses toward conspecific signals over heterospecific signals, and toward particular features of conspecific signals, lies in the tuning of the peripheral auditory system. Biases in processing signals are apparent through the brainstem auditory system, where additional feature detection neurons are added by the time processing reaches the level of the midbrain. Recent work using immediate early gene expression as a marker of neural activity suggests that by the level of the midbrain and forebrain, the differential neural representation of conspecific and heterospecific signals involves both changes in mean activity levels across multiple subnuclei, and in the functional correlations among acoustically active areas. Our data show that in frogs the auditory midbrain appears to play an important role in controlling behavioral responses to acoustic social signals by acting as a regulatory gateway between the stimulus analysis of the brainstem and the behavioral and physiological control centers of the forebrain. We predict that this will hold true for other vertebrate groups such as birds and fish that produce acoustic social signals, and perhaps also in fish where electroreception or vibratory sensing through the lateral line systems plays a role in social signaling, as in all these cases ascending sensory information converges onto midbrain nuclei which relay information to higher brain centers.
Copyright © 2010 Elsevier Ltd. All rights reserved.
Figures

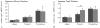

Similar articles
-
Brain Activation Patterns in Response to Conspecific and Heterospecific Social Acoustic Signals in Female Plainfin Midshipman Fish, Porichthys notatus.Brain Behav Evol. 2018;91(1):31-44. doi: 10.1159/000487122. Epub 2018 Mar 29. Brain Behav Evol. 2018. PMID: 29597197 Free PMC article.
-
Sound source perception in anuran amphibians.Curr Opin Neurobiol. 2012 Apr;22(2):301-10. doi: 10.1016/j.conb.2011.12.014. Epub 2012 Jan 20. Curr Opin Neurobiol. 2012. PMID: 22265243 Free PMC article. Review.
-
Influence of descending forebrain projections on processing of acoustic signals and audiomotor integration in the anuran midbrain.Eur J Morphol. 1999 Apr;37(2-3):182-4. doi: 10.1076/ejom.37.2.182.4753. Eur J Morphol. 1999. PMID: 10342453
-
Functional mapping of the auditory midbrain during mate call reception.J Neurosci. 2004 Dec 15;24(50):11264-72. doi: 10.1523/JNEUROSCI.2079-04.2004. J Neurosci. 2004. PMID: 15601932 Free PMC article.
-
Neurobiology of Female Mate Choice in Frogs: Auditory Filtering and Valuation.Integr Comp Biol. 2017 Oct 1;57(4):857-864. doi: 10.1093/icb/icx098. Integr Comp Biol. 2017. PMID: 29048536 Review.
Cited by
-
Auditory brainstem responses in Cope's gray treefrog (Hyla chrysoscelis): effects of frequency, level, sex and size.J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2014 Mar;200(3):221-38. doi: 10.1007/s00359-014-0880-8. Epub 2014 Jan 18. J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2014. PMID: 24442647 Free PMC article.
-
Assessing stimulus and subject influences on auditory evoked potentials and their relation to peripheral physiology in green treefrogs (Hyla cinerea).Comp Biochem Physiol A Mol Integr Physiol. 2014 Dec;178:68-81. doi: 10.1016/j.cbpa.2014.08.005. Epub 2014 Aug 20. Comp Biochem Physiol A Mol Integr Physiol. 2014. PMID: 25151643 Free PMC article.
-
Neuronal Organization in the Inferior Colliculus Revisited with Cell-Type-Dependent Monosynaptic Tracing.J Neurosci. 2018 Mar 28;38(13):3318-3332. doi: 10.1523/JNEUROSCI.2173-17.2018. Epub 2018 Feb 26. J Neurosci. 2018. PMID: 29483283 Free PMC article.
-
Chronic recording of brain activity in awake toads.bioRxiv [Preprint]. 2024 Oct 18:2024.10.16.618567. doi: 10.1101/2024.10.16.618567. bioRxiv. 2024. Update in: J Neurosci Methods. 2025 Jul;419:110449. doi: 10.1016/j.jneumeth.2025.110449. PMID: 39463987 Free PMC article. Updated. Preprint.
-
Inherent Directionality Determines Spatial Release from Masking at the Tympanum in a Vertebrate with Internally Coupled Ears.J Assoc Res Otolaryngol. 2016 Aug;17(4):259-70. doi: 10.1007/s10162-016-0568-6. Epub 2016 Apr 28. J Assoc Res Otolaryngol. 2016. PMID: 27125545 Free PMC article.
References
-
- Fritzsch B, Ryan MJ, Wilczynski W, Hetherington TE, Walkowiak W, editors. The Evoution of the Amphibian Auditory System. New York: Wiley; 1988.
-
- Narins PN, Feng AS, Fay RR, Popper AN, editors. Hearing and Sound Communication in Amphibians. New York: Springer-Verlag; 2007.
-
- Ryan MJ, editor. Anuran Communication. Washington DC: Smithsonian Institution Press; 2001.
-
- Wells KD. The social behavior of anuran amphibians. Animal Behaviour. 1977;25:666–693. Well’s paper is an excellent, comprehensive review of the literature on anuran acoustic communication up to its publication date. It is widely considered a starting point for background information and general overview of anuran acoustic communication across species, including male signaling and both male and female responses.
-
- Ryan MJ, Bernal XE, Rand AS. Female mate choice and the potential for ornament evolution in túngara frogs. Physalaemus pustulosus Current Zoology. 2010;56:343–357. Ryan et al. review experimental studies of female mate choice in túngara frogs to examine an important general issue in evolutionary animal behavior, the evolution elaborate male signals. The authors show that a variety of additions to the conspecific mate recognition signal, both naturally occurring and artificial, enhance the attractiveness of the call to females. The authors argue that latent female preferences for a variety of signal elaborations or innovations can drive the evolution of male signals.
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

