Nocturnal Dexamethasone versus Hydrocortisone for the Treatment of Children with Congenital Adrenal Hyperplasia
- PMID: 20871859
- PMCID: PMC2943098
- DOI: 10.1155/2010/347636
Nocturnal Dexamethasone versus Hydrocortisone for the Treatment of Children with Congenital Adrenal Hyperplasia
Abstract
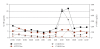
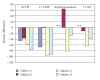
Classic congenital adrenal hyperplasia affects approximately 1 in 15,000 children. Current treatment strategies using multiple daily doses of hydrocortisone lead to suboptimal outcomes. We tested the hypothesis that nocturnal administration of dexamethasone will suppress the hypothalamic-pituitary-adrenal axis more effectively than standard hydrocortisone treatment by blocking the inherent diurnal secretion of ACTH. We performed a pilot study of five prepubertal patients comparing CAH control during two 24-hour hospitalizations, one on hydrocortisone and the other on dexamethasone. The patterns of adrenal suppression differed markedly between hydrocortisone and nocturnal dexamethasone, with significant suppression of the morning rise in ACTH, 17-hydroxyprogesterone, and androstenedione while on dexamethasone. On hydrocortisone therapy, there is a marked variation in ACTH and adrenal hormones depending on time of day and timing of hydrocortisone administration. Longer-term studies are needed to investigate the lowest effective dose and potential toxicity of nocturnal dexamethasone to determine its utility as a therapy for CAH.
Figures

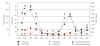


Similar articles
-
Differential effects of hydrocortisone, prednisone, and dexamethasone on hormonal and pharmacokinetic profiles: a pilot study in children with congenital adrenal hyperplasia.Int J Pediatr Endocrinol. 2016;2016:17. doi: 10.1186/s13633-016-0035-5. Epub 2016 Sep 26. Int J Pediatr Endocrinol. 2016. PMID: 27688786 Free PMC article.
-
Serum cortisol and 17-hydroxyprogesterone interrelation in classic 21-hydroxylase deficiency: is current replacement therapy satisfactory?J Clin Endocrinol Metab. 2001 Oct;86(10):4679-85. doi: 10.1210/jcem.86.10.7972. J Clin Endocrinol Metab. 2001. PMID: 11600525
-
Oral hydrocortisone administration in children with classic 21-hydroxylase deficiency leads to more synchronous joint GH and cortisol secretion.J Clin Endocrinol Metab. 2002 May;87(5):2238-44. doi: 10.1210/jcem.87.5.8503. J Clin Endocrinol Metab. 2002. PMID: 11994370
-
PERSPECTIVE: Treatment with hydrocortisone modified-release capsules in children and adolescents with congenital adrenal hyperplasia: an expert opinion.Endocr Connect. 2025 Mar 26;14(5):e240619. doi: 10.1530/EC-24-0619. Print 2025 May 1. Endocr Connect. 2025. PMID: 40094399 Free PMC article. Review.
-
Adrenal disease in pregnancy.Best Pract Res Clin Endocrinol Metab. 2011 Dec;25(6):959-73. doi: 10.1016/j.beem.2011.08.004. Best Pract Res Clin Endocrinol Metab. 2011. PMID: 22115169 Review.
Cited by
-
Novel insights into glucocorticoid replacement therapy for pediatric and adult adrenal insufficiency.Ther Adv Endocrinol Metab. 2019 Feb 2;10:2042018818821294. doi: 10.1177/2042018818821294. eCollection 2019. Ther Adv Endocrinol Metab. 2019. PMID: 30746120 Free PMC article. Review.
-
Rapid hypothalamic-pituitary recovery after chronic glucocorticoid therapy enables strategies that prevent adrenal suppression.bioRxiv [Preprint]. 2025 May 4:2025.04.30.651350. doi: 10.1101/2025.04.30.651350. bioRxiv. 2025. PMID: 40655007 Free PMC article. Preprint.
-
Differential effects of hydrocortisone, prednisone, and dexamethasone on hormonal and pharmacokinetic profiles: a pilot study in children with congenital adrenal hyperplasia.Int J Pediatr Endocrinol. 2016;2016:17. doi: 10.1186/s13633-016-0035-5. Epub 2016 Sep 26. Int J Pediatr Endocrinol. 2016. PMID: 27688786 Free PMC article.
-
Variation in serum adrenal hormones in female 21-hydroxylase deficient patients.Endocr Connect. 2022 Jun 29;11(7):e220143. doi: 10.1530/EC-22-0143. Print 2022 Jul 1. Endocr Connect. 2022. PMID: 35904224 Free PMC article.
-
Hormonal circadian rhythms in patients with congenital adrenal hyperplasia: identifying optimal monitoring times and novel disease biomarkers.Eur J Endocrinol. 2015 Dec;173(6):727-37. doi: 10.1530/EJE-15-0064. Epub 2015 Sep 4. Eur J Endocrinol. 2015. PMID: 26340969 Free PMC article.
References
-
- Cutler GB, Jr., Laue L. Congenital adrenal hyperplasia due to 21-hydroxylase deficiency. The New England Journal of Medicine. 1990;323(26):1806–1813. - PubMed
-
- Bonfig W, Pozza SBD, Schmidt H, Pagel P, Knorr D, Schwarz HP. Hydrocortisone dosing during puberty in patients with classical congenital adrenal hyperplasia: an evidence-based recommendation. Journal of Clinical Endocrinology and Metabolism. 2009;94(10):3882–3888. - PubMed
-
- Eugster EA, DiMeglio LA, Wright JC, Freidenberg GR, Seshadri R, Pescovitz OH. Height outcome in congenital adrenal hyperplasia caused by 21-hydroxylase deficiency: a meta-analysis. Journal of Pediatrics. 2001;138(1):26–32. - PubMed
-
- Stewart PM. The adrenal cortex. In: Melmed S, Kronenberg H, Polonsky K, Larsen PR, editors. Williams Textbook of Endocrinology. Philadelphia, Pa, USA: Saunders Elsevier; 2008. pp. 445–504.
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

