β-nicotinamide adenine dinucleotide is an enteric inhibitory neurotransmitter in human and nonhuman primate colons
- PMID: 20875415
- PMCID: PMC3031738
- DOI: 10.1053/j.gastro.2010.09.039
β-nicotinamide adenine dinucleotide is an enteric inhibitory neurotransmitter in human and nonhuman primate colons
Abstract
Background & aims: An important component of enteric inhibitory neurotransmission is mediated by a purine neurotransmitter, such as adenosine 5'-triphosphate (ATP), binding to P2Y1 receptors and activating small conductance K(+) channels. In murine colon β-nicotinamide adenine dinucleotide (β-NAD) is released with ATP and mimics the pharmacology of inhibitory neurotransmission better than ATP. Here β-NAD and ATP were compared as possible inhibitory neurotransmitters in human and monkey colons.
Methods: A small-volume superfusion assay and high-pressure liquid chromatography with fluorescence detection were used to evaluate spontaneous and nerve-evoked overflow of β-NAD, ATP, and metabolites. Postjunctional responses to nerve stimulation, β-NAD and ATP were compared using intracellular membrane potential and force measurements. Effects of β-NAD on smooth muscle cells (SMCs) were recorded by patch clamp. P2Y receptor transcripts were assayed by reverse transcription polymerase chain reaction.
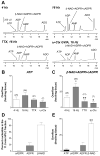
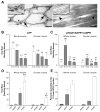
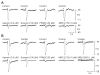
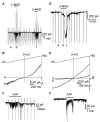
Results: In contrast to ATP, overflow of β-NAD evoked by electrical field stimulation correlated with stimulation frequency and was diminished by the neurotoxins, tetrodotoxin, and ω-conotoxin GVIA. Inhibitory junction potentials and responses to exogenous β-NAD, but not ATP, were blocked by P2Y receptor antagonists suramin, pyridoxal-phosphate-6-azophenyl-2',4'-disulfonate (PPADS), 2'-deoxy-N6-methyladenosine 3',5'-bisphosphate (MRS 2179), and (1R,2S,4S,5S)-4-[2-Iodo-6-(methylamino)-9H-purin-9-yl]-2-(phosphonooxy)bicyclo[3.1.0]hexane-1-methanol dihydrogen phosphate ester tetraammonium salt (MRS 2500). β-NAD activated nonselective cation currents in SMCs, but failed to activate outward currents.
Conclusions: β-NAD meets the criteria for a neurotransmitter better than ATP in human and monkey colons and therefore may contribute to neural regulation of colonic motility. SMCs are unlikely targets for inhibitory purine neurotransmitters because dominant responses of SMCs were activation of net inward, rather than outward, current.
Copyright © 2011. Published by Elsevier Inc.
Conflict of interest statement
Figures


Comment in
-
A new player in neuromuscular transmission in the gastrointestinal tract.Gastroenterology. 2011 Feb;140(2):397-400. doi: 10.1053/j.gastro.2010.12.016. Epub 2010 Dec 18. Gastroenterology. 2011. PMID: 21172334 No abstract available.
-
Evidence for β-nicotinamide adenine dinucleotide as a purinergic, inhibitory neurotransmitter in doubt.Gastroenterology. 2011 Oct;141(4):e27; author reply e27-8. doi: 10.1053/j.gastro.2011.07.047. Epub 2011 Sep 6. Gastroenterology. 2011. PMID: 21888906 No abstract available.
References
-
- Kunze WA, Furness JB. The enteric nervous system and regulation of intestinal motility. Annu Rev Physiol. 1999;61:117–42. - PubMed
-
- Camilleri M, Cowen T, Koch TR. Enteric neurodegeneration in ageing. Neurogastroenterol Motil. 2008;20:418–429. - PubMed
-
- Lomax AE, Sharkey KA, Furness JB. The participation of the sympathetic innervation of the gastrointestinal tract in disease states. Neurogastroenterol Motil. 2010;22:7–18. - PubMed
-
- Wood JD. The enteric purinergic P2Y1 receptor. Curr Opin Pharmacol. 2006;6:564–570. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

