TGF-β2 suppresses macrophage cytokine production and mucosal inflammatory responses in the developing intestine
- PMID: 20875417
- PMCID: PMC3008335
- DOI: 10.1053/j.gastro.2010.09.043
TGF-β2 suppresses macrophage cytokine production and mucosal inflammatory responses in the developing intestine
Abstract
Background & aims: Premature neonates are predisposed to necrotizing enterocolitis (NEC), an idiopathic, inflammatory bowel necrosis. We investigated whether NEC occurs in the preterm intestine due to incomplete noninflammatory differentiation of intestinal macrophages, which increases the risk of a severe mucosal inflammatory response to bacterial products.
Methods: We compared inflammatory properties of human/murine fetal, neonatal, and adult intestinal macrophages. To investigate gut-specific macrophage differentiation, we next treated monocyte-derived macrophages with conditioned media from explanted human fetal and adult intestinal tissues. Transforming growth factor-β (TGF-β) expression and bioactivity were measured in fetal/adult intestine and in NEC. Finally, we used wild-type and transgenic mice to investigate the effects of deficient TGF-β signaling on NEC-like inflammatory mucosal injury.
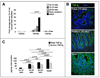
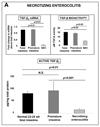
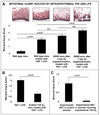
Results: Intestinal macrophages in the human preterm intestine (fetus/premature neonate), but not in full-term neonates and adults, expressed inflammatory cytokines. Macrophage cytokine production was suppressed in the developing intestine by TGF-β, particularly the TGF-β(2) isoform. NEC was associated with decreased tissue expression of TGF-β(2) and decreased TGF-β bioactivity. In mice, disruption of TGF-β signaling worsened NEC-like inflammatory mucosal injury, whereas enteral supplementation with recombinant TGF-β(2) was protective.
Conclusions: Intestinal macrophages progressively acquire a noninflammatory profile during gestational development. TGF-β, particularly the TGF-β(2) isoform, suppresses macrophage inflammatory responses in the developing intestine and protects against inflammatory mucosal injury. Enterally administered TGF-β(2) protected mice from experimental NEC-like injury.
Copyright © 2011 AGA Institute. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Figures






References
-
- Bury RG, Tudehope D. Enteral antibiotics for preventing necrotizing enterocolitis in low birthweight or preterm infants. Cochrane Database Syst Rev. 2001 CD000405. - PubMed
-
- Smythies LE, Maheshwari A, Clements R, Eckhoff D, Novak L, Vu HL, Mosteller-Barnum LM, Sellers M, Smith PD. Mucosal IL-8 and TGF-beta recruit blood monocytes: evidence for cross-talk between the lamina propria stroma and myeloid cells. J Leukoc Biol. 2006;80:492–496. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- HD043397/HD/NICHD NIH HHS/United States
- K08 HD046513/HD/NICHD NIH HHS/United States
- C06RR15490/RR/NCRR NIH HHS/United States
- DK64400/DK/NIDDK NIH HHS/United States
- R24 DK064400/DK/NIDDK NIH HHS/United States
- K12 HD043397/HD/NICHD NIH HHS/United States
- HL092906/HL/NHLBI NIH HHS/United States
- R01 ES015323/ES/NIEHS NIH HHS/United States
- ES015323/ES/NIEHS NIH HHS/United States
- ATS PH-06-006/PH/PHPPO CDC HHS/United States
- C06 RR015490/RR/NCRR NIH HHS/United States
- R01 HD059142/HD/NICHD NIH HHS/United States
- R01 HL092906/HL/NHLBI NIH HHS/United States
- HD046513/HD/NICHD NIH HHS/United States
- HD059142/HD/NICHD NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

