Comparative analysis of immune responses to Russian spring-summer encephalitis and Omsk hemorrhagic fever viruses in mouse models
- PMID: 20875909
- PMCID: PMC2966520
- DOI: 10.1016/j.virol.2010.08.021
Comparative analysis of immune responses to Russian spring-summer encephalitis and Omsk hemorrhagic fever viruses in mouse models
Abstract
Omsk hemorrhagic fever virus (OHFV) and Russian spring-summer encephalitis virus (RSSEV) are tick-borne flaviviruses that have close homology but different pathology and disease outcomes. Previously, we reported that C57BL/6 and BALB/c mice were excellent models to study the pathology and clinical signs of human RSSEV and OHFV infection. In the study described here, we found that RSSEV infection induced robust release of proinflammatory cytokines (IL-1α, IL-1β, IL-6 and TNF-α) and chemokines (MCP-1, MIP-1β, RANTES and KC) in the brain at 9 and 11dpi, together with moderate to low Th1 and Th2 cytokines. In contrast, OHFV infection stimulated an early and prominent induction of IL-1α, TNF-α, IL-12p70, MCP-1, MIP-1α and MIP-1β in the spleen of infected mice. Collectively our data suggest that a differential host response to infection may lead to the alternate disease outcomes seen following OHFV or RSSEV infection.
Copyright © 2010 Elsevier Inc. All rights reserved.
Figures
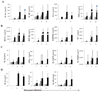
 ) mock, (
) mock, ( ) OHFV or (
) OHFV or ( ) RSSEV infection. Protein levels of (A) proinflammatory cytokines, (B) Chemokines, (C) Th1 cytokines and (D) Th2 cytokines were measured in the brain at the indicated time points following foot pad injection of 1000pfu OHFV or RSSEV. Data are shown as mean ± SD for 3–5 mice at each time point. *p<0.05 for the comparison between RSSEV and mock,
) RSSEV infection. Protein levels of (A) proinflammatory cytokines, (B) Chemokines, (C) Th1 cytokines and (D) Th2 cytokines were measured in the brain at the indicated time points following foot pad injection of 1000pfu OHFV or RSSEV. Data are shown as mean ± SD for 3–5 mice at each time point. *p<0.05 for the comparison between RSSEV and mock,  p<0.05 for comparison between RSSEV and OHFV.
p<0.05 for comparison between RSSEV and OHFV.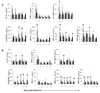
 ) mock, (
) mock, ( ) OHFV or (
) OHFV or ( ) RSSEV infection of (A) BALB/c and (B) C57BL/6 mice. Spleen was extracted at the indicated time and the level of the cytokine was measured from the clear homogenates. Data are the mean ± SD for 3–5 mice at each time point. *p<0.05 for the comparison between OHFV/RSSEV and mock,
) RSSEV infection of (A) BALB/c and (B) C57BL/6 mice. Spleen was extracted at the indicated time and the level of the cytokine was measured from the clear homogenates. Data are the mean ± SD for 3–5 mice at each time point. *p<0.05 for the comparison between OHFV/RSSEV and mock,  p<0.05 for comparison between RSSEV and OHFV.
p<0.05 for comparison between RSSEV and OHFV.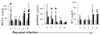
 ) mock, (
) mock, ( ) OHFV or (
) OHFV or ( ) RSSEV infection of BALB/ c mice. Lung and liver were extracted at the indicated days and the supernatant was used to analyze the level of cytokines/chemokines. Data are shown as mean ± SD for 3–5 mice at each time point. *p<0.05 for the comparison between OHFV/RSSEV and mock,
) RSSEV infection of BALB/ c mice. Lung and liver were extracted at the indicated days and the supernatant was used to analyze the level of cytokines/chemokines. Data are shown as mean ± SD for 3–5 mice at each time point. *p<0.05 for the comparison between OHFV/RSSEV and mock,  p<0.05 for comparison between RSSEV and OHFV.
p<0.05 for comparison between RSSEV and OHFV.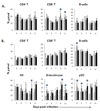
 ) mock, (
) mock, ( ) OHFV or (
) OHFV or ( ) RSSEV. Dissociated splenocytes were isolated on the designated days post infection. The cells were stained with appropriate antibodies and analyzed using flow cytometry. Data are shown as mean ± SD for 5 mice at each time point. *p<0.05 for the comparison between OHFV/RSSEV and mock,
) RSSEV. Dissociated splenocytes were isolated on the designated days post infection. The cells were stained with appropriate antibodies and analyzed using flow cytometry. Data are shown as mean ± SD for 5 mice at each time point. *p<0.05 for the comparison between OHFV/RSSEV and mock,  p<0.05 for comparison between RSSEV and OHFV.
p<0.05 for comparison between RSSEV and OHFV.Similar articles
-
An animal model for the tickborne flavivirus--Omsk hemorrhagic fever virus.J Infect Dis. 2005 Jan 1;191(1):100-8. doi: 10.1086/426397. Epub 2004 Dec 3. J Infect Dis. 2005. PMID: 15593010
-
Evaluation of the European tick-borne encephalitis vaccine against Omsk hemorrhagic fever virus.Microbiol Immunol. 2014 Feb;58(2):112-8. doi: 10.1111/1348-0421.12122. Microbiol Immunol. 2014. PMID: 24329534
-
Cross-Reactive Antibodies to the NS1 Protein of Omsk Hemorrhagic Fever Virus Are Absent in the Sera of Patients with Tick-Borne Encephalitis.Viruses. 2024 Jun 27;16(7):1032. doi: 10.3390/v16071032. Viruses. 2024. PMID: 39066195 Free PMC article.
-
[Tick-borne human pathogenic microorganisms found in Europe and those considered nonpathogenic. Part I: Ticks and Viruses].Bundesgesundheitsblatt Gesundheitsforschung Gesundheitsschutz. 2004 Apr;47(4):392-404. doi: 10.1007/s00103-003-0766-3. Bundesgesundheitsblatt Gesundheitsforschung Gesundheitsschutz. 2004. PMID: 15205783 Review. German.
-
Omsk haemorrhagic fever.Lancet. 2010 Dec 18;376(9758):2104-13. doi: 10.1016/S0140-6736(10)61120-8. Epub 2010 Sep 16. Lancet. 2010. PMID: 20850178 Review.
Cited by
-
Comparative pathogenesis of Alkhumra hemorrhagic fever and Kyasanur forest disease viruses in a mouse model.PLoS Negl Trop Dis. 2014 Jun 12;8(6):e2934. doi: 10.1371/journal.pntd.0002934. eCollection 2014 Jun. PLoS Negl Trop Dis. 2014. PMID: 24922308 Free PMC article.
-
Lethal Experimental Tick-Borne Encephalitis Infection: Influence of Two Strains with Similar Virulence on the Immune Response.Front Microbiol. 2017 Jan 20;7:2172. doi: 10.3389/fmicb.2016.02172. eCollection 2016. Front Microbiol. 2017. PMID: 28163697 Free PMC article.
-
Tick-borne encephalitis virus induces chemokine RANTES expression via activation of IRF-3 pathway.J Neuroinflammation. 2016 Aug 30;13(1):209. doi: 10.1186/s12974-016-0665-9. J Neuroinflammation. 2016. PMID: 27576490 Free PMC article.
-
Chemokine receptors as important regulators of pathogenesis during arboviral encephalitis.Front Cell Neurosci. 2014 Sep 30;8:264. doi: 10.3389/fncel.2014.00264. eCollection 2014. Front Cell Neurosci. 2014. PMID: 25324719 Free PMC article. Review.
-
A database of human genes and a gene network involved in response to tick-borne encephalitis virus infection.BMC Evol Biol. 2017 Dec 28;17(Suppl 2):259. doi: 10.1186/s12862-017-1107-8. BMC Evol Biol. 2017. PMID: 29297316 Free PMC article.
References
-
- Biswas SM, Kar S, Singh R, Chakraborty D, Vipat V, Raut CG, Mishra AC, Gore MM, Ghosh D. Immunomodulatory cytokines determine the outcome of Japanese encephalitis virus infection in mice. J. Med. Virol. 2010;82:304–310. - PubMed
-
- Chen CJ, Liao SL, Kuo MD, Wang YM. Astrocytic alteration induced by Japanese encephalitis virus infection. Neuroreport. 2000;11:1933–1937. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

