Angio-adaptation in unloaded skeletal muscle: new insights into an early and muscle type-specific dynamic process
- PMID: 20876198
- PMCID: PMC3008859
- DOI: 10.1113/jphysiol.2010.193243
Angio-adaptation in unloaded skeletal muscle: new insights into an early and muscle type-specific dynamic process
Abstract
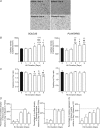
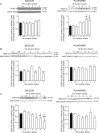
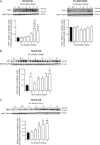
With a remarkable plasticity, skeletal muscle adapts to an altered functional demand. Muscle angio-adaptation can either involve the growth or the regression of capillaries as respectively observed in response to endurance training or muscle unloading. Whereas the molecular mechanisms that regulate exercise-induced muscle angiogenesis have been extensively studied, understanding how muscle unloading can in contrast lead to capillary regression has received very little attention. Here we have investigated the consequences of a 9 day time course hindlimb unloading on both capillarization and expression of angio-adaptive molecules in two different rat skeletal muscles. Both soleus and plantaris muscles were atrophied similarly. In contrast, our results have shown different angio-adaptive patterns between these two muscles. Capillary regression occurred only in the soleus, a slow-twitch and oxidative postural muscle. Conversely, the level of capillarization was preserved in the plantaris, a fast-twitch and glycolytic muscle. We have also measured the time course protein expression of key pro- and anti-angiogenic signals (VEGF-A, VEGF-B, VEGF-R2, TSP-1). Our results have revealed that the angio-adaptive response to unloading was muscle-type specific, and that an integrated balance between pro- and anti-angiogenic signals plays a determinant role in regulating this process. In conclusion, we have brought new evidence that measuring the ratio between pro- and anti-angiogenic signals in order to evaluate muscle angio-adaptation was a more accurate approach than analysing the expression of molecular factors taken individually.
Figures
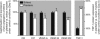

References
-
- Alway SE, Lowe DA, Chen KD. The effects of age and hindlimb supension on the levels of expression of the myogenic regulatory factors MyoD and myogenin in rat fast and slow skeletal muscles. Exp Physiol. 2001;86:509–517. - PubMed
-
- An WG, Kanekal M, Simon MC, Maltepe E, Blagosklonny MV, Neckers LM. Stabilization of wild-type p53 by hypoxia-inducible factor 1α. Nature. 1998;392:405–408. - PubMed
-
- Bellomo D, Headrick JP, Silins GU, Paterson CA, Thomas PS, Gartside M, Mould A, Cahill MM, Tonks ID, Grimmond SM, Townson S, Wells C, Little M, Cummings MC, Hayward NK, Kay GF. Mice lacking the vascular endothelial growth factor-B gene (Vegfb) have smaller hearts, dysfunctional coronary vasculature, and impaired recovery from cardiac ischemia. Circ Res. 2000;86:E29–35. - PubMed
-
- Blagosklonny MV, Giannakakou P, Wojtowicz M, Romanova LY, Ain KB, Bates SE, Fojo T. Effects of p53-expressing adenovirus on the chemosensitivity and differentiation of anaplastic thyroid cancer cells. J Clin Endocrinol Metab. 1998;83:2516–2522. - PubMed
-
- Booth FW, Thomason DB. Molecular and cellular adaptation of muscle in response to exercise: perspectives of various models. Physiol Rev. 1991;71:541–585. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Miscellaneous

