Lactate per se improves the excitability of depolarized rat skeletal muscle by reducing the Cl- conductance
- PMID: 20876199
- PMCID: PMC3010146
- DOI: 10.1113/jphysiol.2010.196568
Lactate per se improves the excitability of depolarized rat skeletal muscle by reducing the Cl- conductance
Abstract
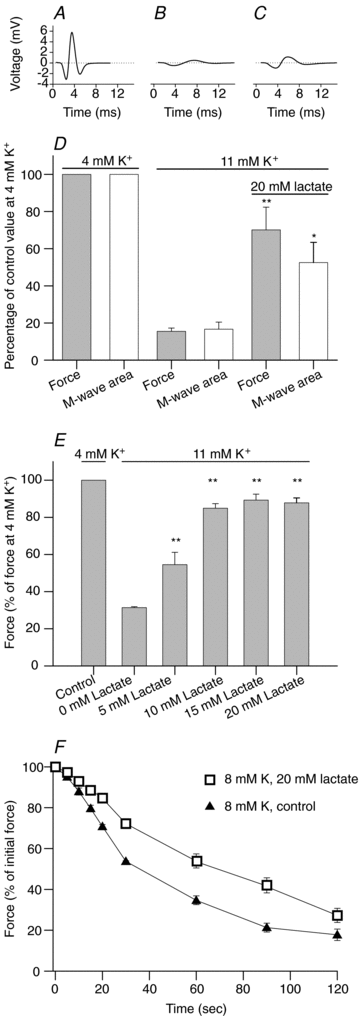
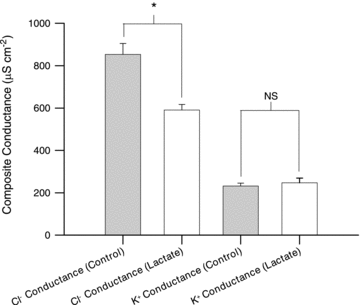
Studies on rats have shown that lactic acid can improve excitability and function of depolarized muscles. The effect has been related to the ensuing reduction in intracellular pH causing inhibition of muscle fibre Cl(-) channels. However, since several carboxylic acids with structural similarities to lactate can inhibit muscle Cl(-) channels it is possible that lactate per se can increase muscle excitability by exerting a direct effect on these channels. We therefore examined the effects of lactate on the function of intact muscles and skinned fibres together with effects on pH and Cl(-) conductance (G(cl)). In muscles where extracellular compound action potentials (M-waves) and tetanic force response to excitation were reduced by (mean ± s.e.m.) 82 ± 4% and 83 ± 2%, respectively, by depolarization with 11 mm extracellular K(+), both M-waves and force exhibited an up to 4-fold increase when 20 mm lactate was added. This effect was present already at 5 mm and saturated at 15 mm lactate, and was associated with a 31% reduction in G(Cl). The effects of lactate were completely blocked by Cl(-) channel inhibition or use of Cl(-)-free solutions. Finally, both experiments where effects of lactate on intracellular pH in intact muscles were mimicked by increased CO₂ tension and experiments with skinned fibres showed that the effects of lactate could not be related to reduced intracellular pH. It is concluded that addition of lactate can inhibit ClC-1 Cl(-) channels and increase the excitability and contractile function of depolarized rat muscles via mechanisms not related to a reduction in intracellular pH.
Figures

References
-
- Andrews MA, Godt RE, Nosek TM. Influence of physiological l(+)-lactate concentrations on contractility of skinned striated muscle fibers of rabbit. J Appl Physiol. 1996;80:2060–2065. - PubMed
-
- Brooks GA. Lactate: link between glycolytic and oxidative metabolism. Sports Med. 2007;37:341–343. - PubMed
-
- Cairns SP. Lactic acid and exercise performance: culprit or friend? Sports Med. 2004;36:279–291. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

