Phase I and pharmacokinetic studies of CYT-6091, a novel PEGylated colloidal gold-rhTNF nanomedicine
- PMID: 20876255
- PMCID: PMC3004980
- DOI: 10.1158/1078-0432.CCR-10-0978
Phase I and pharmacokinetic studies of CYT-6091, a novel PEGylated colloidal gold-rhTNF nanomedicine
Abstract
Purpose: A novel nanomedicine, CYT-6091, constructed by simultaneously binding recombinant human tumor necrosis factor alpha (rhTNF) and thiolyated polyethylene glycol to the surface of 27-nm colloidal gold particles, was tested in a phase I dose escalation clinical trial in advanced stage cancer patients.
Experimental design: CYT-6091, whose dosing was based on the amount of rhTNF in the nanomedicine, was injected intravenously, and 1 cycle of treatment consisted of 2 treatments administered 14 days apart.
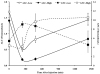
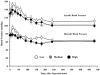
Results: Doses from 50 μg/m(2) to 600 μg/m(2) were well tolerated, and no maximum tolerated dose (MTD) was reached, as the highest dose exceeded the target dosage of 1-mg rhTNF per treatment, exceeding the previous MTD for native rhTNF by 3-fold. The first 2 patients on the study, each receiving 50 μg/m(2), did not receive any prophylactic antipyretics or H2 blockade. A predicted, yet controllable fever occurred in these patients, so all subsequently treated patients received prophylactic antipyretics and H2 blockers. However, even at the highest dose rhTNF's dose-limiting toxic effect of hypotension was not seen. Using electron microscopy to visualize nanoparticles of gold in patient biopsies of tumor and healthy tissue showed that patient biopsies taken 24 hours after treatment had nanoparticles of gold in tumor tissue.
Conclusions: These data indicate that rhTNF formulated as CYT-6091 may be administered systemically at doses of rhTNF that were previously shown to be toxic and that CYT-6091 may target to tumors. Future clinical studies will focus on combining CYT-6091 with approved chemotherapies for the systemic treatment of nonresectable cancers.
©2010 AACR.
Figures

References
-
- Alexander HR, Feldman AL. Tumor necrosis factor: Basic principles and clinical application in systemic and regional cancer treatment. In: Steven A, Rosenberg MD, editors. Biologic Therapy of Cancer. 3. Philadelphia: Lippincott; 2000. pp. 174–93.
-
- Kimura K, Taguchi T, Urushizaki I, Ohno R, Abe O, Furue H, et al. Phase I study of recombinant human tumor necrosis factor. Cancer Chemother Pharmacol. 1987;20:223–229. - PubMed
-
- Taguchi T. Phase I study of recombinant human tumor necrosis factor (rHu-TNF:PT-050) Cancer Detection and Prevention. 1988;12:561–572. - PubMed
-
- Creaven PJ, Plager JE, Dupere S, Huben RP, Takita H, Mittelman A, et al. Phase I clinical trial of recombinant human tumor necrosis factor. Cancer Chemotherapy and Pharmacology. 1987;20:137–144. - PubMed
-
- Tracey KJ, Beutler B, Lowry SF, Merryweather J, Wolpe S, Milsark IW, et al. Shock and tissue injury induced by recombinant human cachectin. Science. 1986;234:470–474. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

