In vitro characterization of the microglial inflammatory response to Streptococcus suis, an important emerging zoonotic agent of meningitis
- PMID: 20876287
- PMCID: PMC2981315
- DOI: 10.1128/IAI.00698-10
In vitro characterization of the microglial inflammatory response to Streptococcus suis, an important emerging zoonotic agent of meningitis
Abstract
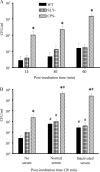
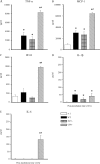
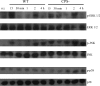
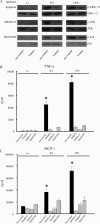
Streptococcus suis is an important swine and human pathogen responsible for septicemia and meningitis. In vivo research in mice suggested that in the brain, microglia might be involved in activating the inflammatory response against S. suis. The aim of this study was to better understand the interactions between S. suis and microglia. Murine microglial cells were infected with a virulent wild-type strain of S. suis. Two isogenic mutants deficient at either capsular polysaccharide (CPS) or hemolysin production were also included. CPS contributed to S. suis resistance to phagocytosis and regulated the inflammatory response by hiding proinflammatory components from the bacterial cell wall, while the absence of hemolysin, a potential cytotoxic factor, did not have a major impact on S. suis interactions with microglia. Wild-type S. suis induced enhanced expression of Toll-like receptor 2 by microglial cells, as well as phosphotyrosine, protein kinase C, and different mitogen-activated protein kinase signaling events. However, cells infected with the CPS-deficient mutant showed overall stronger and more sustained phosphorylation profiles. CPS also modulated inducible nitric oxide synthase expression and further nitric oxide production from S. suis-infected microglia. Finally, S. suis-induced NF-κB translocation was faster for cells stimulated with the CPS-deficient mutant, suggesting that bacterial cell wall components are potent inducers of NF-κB. These results contribute to increase the knowledge of mechanisms underlying S. suis inflammation in the brain and will be useful in designing more efficient anti-inflammatory strategies for meningitis.
Figures





Similar articles
-
Autophagy is a defense mechanism controlling Streptococcus suis serotype 2 infection in murine microglia cells.Vet Microbiol. 2021 Jul;258:109103. doi: 10.1016/j.vetmic.2021.109103. Epub 2021 May 9. Vet Microbiol. 2021. PMID: 33991788
-
Autophagy inhibits nuclear factor kappa B and mitogen-activated protein kinase (MAPK) inflammatory signaling pathways and modulates cytokine release in murine microglia following Streptococcus suis serotype 2 infection.J Vet Med Sci. 2025 Jan 10;87(1):68-74. doi: 10.1292/jvms.24-0203. Epub 2024 Nov 26. J Vet Med Sci. 2025. PMID: 39603605 Free PMC article.
-
Streptococcus suis: a re-emerging pathogen associated with occupational exposure to pigs or pork products. Part II - Pathogenesis.Ann Agric Environ Med. 2018 Mar 14;25(1):186-203. doi: 10.26444/aaem/85651. Epub 2018 Mar 2. Ann Agric Environ Med. 2018. PMID: 29575852 Review.
-
Evaluation of the pathogenesis of meningitis caused by Streptococcus suis sequence type 7 using the infection of BV2 microglial cells.J Med Microbiol. 2013 Mar;62(Pt 3):360-368. doi: 10.1099/jmm.0.046698-0. Epub 2012 Dec 6. J Med Microbiol. 2013. PMID: 23222863
-
Streptococcus suis infection: an emerging/reemerging challenge of bacterial infectious diseases?Virulence. 2014 May 15;5(4):477-97. doi: 10.4161/viru.28595. Epub 2014 Mar 25. Virulence. 2014. PMID: 24667807 Free PMC article. Review.
Cited by
-
Toll-like receptor 2-independent host innate immune response against an epidemic strain of Streptococcus suis that causes a toxic shock-like syndrome in humans.PLoS One. 2013 May 28;8(5):e65031. doi: 10.1371/journal.pone.0065031. Print 2013. PLoS One. 2013. PMID: 23724118 Free PMC article.
-
Toll-like receptor 2 ligand pretreatment attenuates retinal microglial inflammatory response but enhances phagocytic activity toward Staphylococcus aureus.Infect Immun. 2012 Jun;80(6):2076-88. doi: 10.1128/IAI.00149-12. Epub 2012 Mar 19. Infect Immun. 2012. PMID: 22431652 Free PMC article.
-
Virulence Studies of Different Sequence Types and Geographical Origins of Streptococcus suis Serotype 2 in a Mouse Model of Infection.Pathogens. 2016 Jul 11;5(3):48. doi: 10.3390/pathogens5030048. Pathogens. 2016. PMID: 27409640 Free PMC article.
-
Interactions of Streptococcus suis serotype 2 with human meningeal cells and astrocytes.BMC Res Notes. 2015 Oct 26;8:607. doi: 10.1186/s13104-015-1581-2. BMC Res Notes. 2015. PMID: 26502903 Free PMC article.
-
Host-pathogen interactions in bacterial meningitis.Acta Neuropathol. 2016 Feb;131(2):185-209. doi: 10.1007/s00401-015-1531-z. Epub 2016 Jan 7. Acta Neuropathol. 2016. PMID: 26744349 Free PMC article. Review.
References
-
- Akira, S., K. Takeda, and T. Kaisho. 2001. Toll-like receptors: critical proteins linking innate and acquired immunity. Nat. Immunol. 2:675-680. - PubMed
-
- Andersen, C. L., J. L. Jensen, and T. F. Ørntoft. 2004. Normalization of real-time quantitative reverse transcription-PCR data: a model-based variance estimation approach to identify genes suited for normalization, applied to bladder and colon cancer data sets. Cancer Res. 64:5245-5250. - PubMed
-
- Bahloul, H., A. Mofredj, A. Mrabet, G. Gineyt, and P. Rousselier. 2008. Streptococcus suis meningitis after oral contamination? Med. Mal. Infect. 38:281-282. - PubMed
-
- Baums, C. G., and P. Valentin-Weigand. 2009. Surface-associated and secreted factors of Streptococcus suis in epidemiology, pathogenesis and vaccine development. Anim. Health Res. Rev. 10:65-83. - PubMed
-
- Benga, L., M. Fulde, C. Neis, R. Goethe, and P. Valentin-Weigand. 2008. Polysaccharide capsule and suilysin contribute to extracellular survival of Streptococcus suis co-cultivated with primary porcine phagocytes. Vet. Microbiol. 132:211-219. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

