Uptake of Helicobacter pylori outer membrane vesicles by gastric epithelial cells
- PMID: 20876296
- PMCID: PMC2981328
- DOI: 10.1128/IAI.00299-10
Uptake of Helicobacter pylori outer membrane vesicles by gastric epithelial cells
Abstract
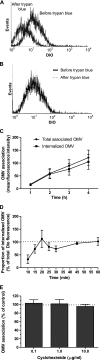
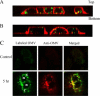
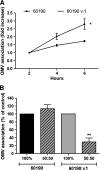
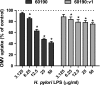
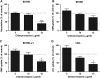
Helicobacter pylori bacteria colonize the human stomach where they stimulate a persistent inflammatory response. H. pylori is considered noninvasive; however, lipopolysaccharide (LPS)-enriched outer membrane vesicles (OMV), continuously shed from the surface of this bacterium, are observed within gastric epithelial cells. The mechanism of vesicle uptake is poorly understood, and this study was undertaken to examine the roles of bacterial VacA cytotoxin and LPS in OMV binding and cholesterol and clathrin-mediated endocytosis in vesicle uptake by gastric epithelial cells. OMV association was examined using a fluorescent membrane dye to label OMV, and a comparison was made between the associations of vesicles from a VacA(+) strain and OMV from a VacA(-) isogenic mutant strain. Within 20 min, essentially all associated OMV were intracellular, and vesicle binding appeared to be facilitated by the presence of VacA cytotoxin. Uptake of vesicles from the VacA(+) strain was inhibited by H. pylori LPS (58% inhibition with 50 μg/ml LPS), while uptake of OMV from the VacA(-) mutant strain was less affected (25% inhibition with 50 μg/ml LPS). Vesicle uptake did not require cholesterol. However, uptake of OMV from the VacA(-) mutant strain was inhibited by a reduction in clathrin-mediated endocytosis (42% with 15 μg/ml chlorpromazine), while uptake of OMV from the VacA(+) strain was less affected (25% inhibition with 15 μg/ml chlorpromazine). We conclude that VacA toxin enhances the association of H. pylori OMV with cells and that the presence of the toxin may allow vesicles to exploit more than one pathway of internalization.
Figures

References
-
- Atherton, J. C., P. Cao, R. M. Peek, Jr., M. K. Tummuru, M. J. Blaser, and T. L. Cover. 1995. Mosaicism in vacuolating cytotoxin alleles of Helicobacter pylori. Association of specific vacA types with cytotoxin production and peptic ulceration. J. Biol. Chem. 270:17771-17777. - PubMed
-
- Ayala, G., L. Torres, M. Espinosa, G. Fierros-Zarate, V. Maldonado, and J. Melendez-Zajgla. 2006. External membrane vesicles from Helicobacter pylori induce apoptosis in gastric epithelial cells. FEMS Microbiol. Lett. 260:178-185. - PubMed
-
- Bayerdorffer, E., A. Neubauer, B. Rudolph, C. Thiede, N. Lehn, S. Eidt, and M. Stolte. 1995. Regression of primary gastric lymphoma of mucosa-associated lymphoid tissue type after cure of Helicobacter pylori infection. MALT Lymphoma Study Group. Lancet 345:1591-1594. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

