Pregabalin for the treatment of men with chronic prostatitis/chronic pelvic pain syndrome: a randomized controlled trial
- PMID: 20876412
- PMCID: PMC3767281
- DOI: 10.1001/archinternmed.2010.319
Pregabalin for the treatment of men with chronic prostatitis/chronic pelvic pain syndrome: a randomized controlled trial
Abstract
Background: Evidence suggests that the urogenital pain of chronic prostatitis/chronic pelvic pain syndrome (CP/CPPS) may be neuropathic.
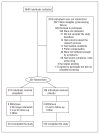
Methods: This randomized, double-blind, placebo-controlled trial was conducted across 10 tertiary care centers in North America to determine whether pregabalin, which has been proved effective in other chronic pain syndromes, is effective in reducing CP/CPPS symptoms. In 2006-2007, 324 men with pelvic pain for at least 3 of the previous 6 months were enrolled in this study. Men were randomly assigned to receive pregabalin or placebo in a 2:1 ratio and were treated for 6 weeks. Pregabalin dosage was increased from 150 to 600 mg/d during the first 4 weeks. The primary outcome was a 6-point decrease in the National Institutes of Health Chronic Prostatitis Symptom Index (NIH-CPSI) total score. Multiple secondary outcomes were assessed.
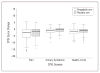
Results: Of 218 men assigned to receive pregabalin, 103 (47.2%) reported at least a 6-point decrease in the NIH-CPSI total score at 6 weeks compared with 35.8% (38 of 106 men) assigned to receive placebo (P = .07, exact Mantel-Haenszel test, adjusting for clinical sites). Compared with the placebo group, men assigned to receive pregabalin experienced reductions in the NIH-CPSI total score and subscores (P < .05), a higher Global Response Assessment response rate (31.2% and 18.9%; P = .02), and improvement in total McGill Pain Questionnaire score (P = .01). Results for the other outcomes did not differ between groups.
Conclusion: Pregabalin therapy for 6 weeks was not superior to placebo use in the rate of a 6-point decrease (improvement) in the NIH-CPSI total score in men with CP/CPPS.
Trial registration: clinicaltrials.gov Identifier: NCT00371033.
Figures
Comment in
-
Re: Pregabalin for the treatment of men with chronic prostatitis/chronic pelvic pain syndrome: a randomized controlled trial.J Urol. 2011 Apr;185(4):1302-3. doi: 10.1016/S0022-5347(11)60282-6. J Urol. 2011. PMID: 22098964 No abstract available.
References
-
- Krieger JN, Nyberg L, Jr, Nickel JC. NIH consensus definition and classification of prostatitis. JAMA. 1999;282(3):236–237. - PubMed
-
- Pontari MA, Joyce GF, Wise M, McNaughton-Collins M Urologic Diseases in America Project. Prostatitis. J Urol. 2007;177(6):2050–2057. - PubMed
-
- Calhoun EA, McNaughton Collins M, Pontari MA, et al. Chronic Prostatitis Collaborative Research Network. The economic impact of chronic prostatitis. Arch Intern Med. 2004;164(11):1231–1236. - PubMed
-
- Pontari MA, McNaughton-Collins M, O'Leary MP, et al. CPCRN Study Group. A case-control study of risk factors in men with chronic pelvic pain syndrome. BJU Int. 2005;96(4):559–565. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
- U01 DK065174/DK/NIDDK NIH HHS/United States
- U01 DK065297/DK/NIDDK NIH HHS/United States
- U01 DK65189/DK/NIDDK NIH HHS/United States
- U01 DK065186/DK/NIDDK NIH HHS/United States
- U01 DK65277/DK/NIDDK NIH HHS/United States
- U01 DK65266/DK/NIDDK NIH HHS/United States
- UO1DK65268/DK/NIDDK NIH HHS/United States
- U01 DK65209/DK/NIDDK NIH HHS/United States
- U01 DK65257/DK/NIDDK NIH HHS/United States
- U01 DK065287/DK/NIDDK NIH HHS/United States
- U01 DK065268/DK/NIDDK NIH HHS/United States
- U01 DK65174/DK/NIDDK NIH HHS/United States
- U01 DK65287/DK/NIDDK NIH HHS/United States
- U01 DK65187/DK/NIDDK NIH HHS/United States
- U01 DK065266/DK/NIDDK NIH HHS/United States
- U01 DK065277/DK/NIDDK NIH HHS/United States
- U01 DK065187/DK/NIDDK NIH HHS/United States
- U01 DK065209/DK/NIDDK NIH HHS/United States
- U01 DK65186/DK/NIDDK NIH HHS/United States
- U01 DK065189/DK/NIDDK NIH HHS/United States
- U01 DK065257/DK/NIDDK NIH HHS/United States
- U01 DK65297/DK/NIDDK NIH HHS/United States
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

