Distinct roles of the ATR kinase and the Mre11-Rad50-Nbs1 complex in the maintenance of chromosomal stability in Arabidopsis
- PMID: 20876831
- PMCID: PMC2965537
- DOI: 10.1105/tpc.110.078527
Distinct roles of the ATR kinase and the Mre11-Rad50-Nbs1 complex in the maintenance of chromosomal stability in Arabidopsis
Abstract
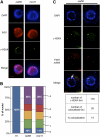
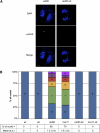
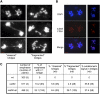
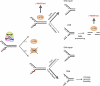
Signaling of chromosomal DNA breaks is of primary importance for initiation of repair and, thus, for global genomic stability. Although the Mre11-Rad50-Nbs1 (MRN) complex is the first sensor of double-strand breaks, its role in double-strand break (DSB) signaling is not fully understood. We report the absence of γ-ray-induced, ATM/ATR-dependent histone H2AX phosphorylation in Arabidopsis thaliana rad50 and mre11 mutants, confirming that the MRN complex is required for H2AX phosphorylation by the ATM and ATR kinases in response to irradiation-induced DSB in Arabidopsis. rad50 and mre11 mutants spontaneously activate a DNA damage response, as shown by the presence of γ-H2AX foci and activation of cell cycle arrest in nonirradiated plants. This response is ATR dependent as shown both by the absence of these spontaneous foci and by the wild-type mitotic indices of double rad50 atr and mre11 atr plants. EdU S-phase labeling and fluorescence in situ hybridization analysis using specific subtelomeric probes point to a replicative S-phase origin of this chromosome damage in the double mutants and not to telomere destabilization. Thus, the data presented here show the exclusive involvement of ATR in DNA damage signaling in MRN mutants and provide evidence for a role for ATR in the avoidance of S-phase DNA damage.
Figures




References
-
- Bleuyard J.-Y., Gallego M.E., White C.I. (2004). Meiotic defects in the Arabidopsis rad50 mutant point to conservation of the MRX complex function in early stages of meiotic recombination. Chromosoma 113: 197–203 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

