Brain water mobility decreases after astrocytic aquaporin-4 inhibition using RNA interference
- PMID: 20877385
- PMCID: PMC3063618
- DOI: 10.1038/jcbfm.2010.163
Brain water mobility decreases after astrocytic aquaporin-4 inhibition using RNA interference
Abstract
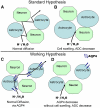
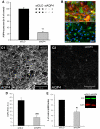
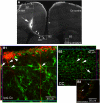
Neuroimaging with diffusion-weighted imaging is routinely used for clinical diagnosis/prognosis. Its quantitative parameter, the apparent diffusion coefficient (ADC), is thought to reflect water mobility in brain tissues. After injury, reduced ADC values are thought to be secondary to decreases in the extracellular space caused by cell swelling. However, the physiological mechanisms associated with such changes remain uncertain. Aquaporins (AQPs) facilitate water diffusion through the plasma membrane and provide a unique opportunity to examine the molecular mechanisms underlying water mobility. Because of this critical role and the recognition that brain AQP4 is distributed within astrocytic cell membranes, we hypothesized that AQP4 contributes to the regulation of water diffusion and variations in its expression would alter ADC values in normal brain. Using RNA interference in the rodent brain, we acutely knocked down AQP4 expression and observed that a 27% AQP4-specific silencing induced a 50% decrease in ADC values, without modification of tissue histology. Our results demonstrate that ADC values in normal brain are modulated by astrocytic AQP4. These findings have major clinical relevance as they suggest that imaging changes seen in acute neurologic disorders such as stroke and trauma are in part due to changes in tissue AQP4 levels.
Figures




References
-
- Amiry-Moghaddam M, Xue R, Haug FM, Neely JD, Bhardwaj A, Agre P, Adams ME, Froehner SC, Mori S, Ottersen OP. Alpha syntrophin deletion removes the perivascular but not the endothelial pool of aquaporin-4 at the blood-brain barrier and delays the development of brain edema in an experimental model of acute hyponatremia. FASEB J. 2004;18:542–544. - PubMed
-
- Auguste KI, Jin S, Uchida K, Yan D, Manley GT, Papadopoulos MC, Verkman AS. Greatly impaired migration of implanted aquaporin-4-deficient astroglial cells in mouse brain toward a site of injury. FASEB J. 2007;21:108–116. - PubMed
-
- Badaut J, Ashwal S, Tone B, Regli L, Tian HR, Obenaus A. Temporal and regional evolution of aquaporin-4 expression and magnetic resonance imaging in a rat pup model of neonatal stroke. Pediatr Res. 2007;62:248–254. - PubMed
-
- Badaut J, Hirt L, Price M, de Castro Ribeiro M, Magistretti PJ, Regli L. Hypoxia/hypoglycemia preconditioning prevents the loss of functional electrical activity in organotypic slice cultures. Brain Res. 2005;1051:117–122. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

