A mouse strain where basal connective tissue growth factor gene expression can be switched from low to high
- PMID: 20877562
- PMCID: PMC2943916
- DOI: 10.1371/journal.pone.0012909
A mouse strain where basal connective tissue growth factor gene expression can be switched from low to high
Abstract
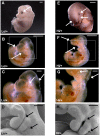
Connective tissue growth factor (CTGF) is a signaling molecule that primarily functions in extracellular matrix maintenance and repair. Increased Ctgf expression is associated with fibrosis in chronic organ injury. Studying the role of CTGF in fibrotic disease in vivo, however, has been hampered by perinatal lethality of the Ctgf null mice as well as the limited scope of previous mouse models of Ctgf overproduction. Here, we devised a new approach and engineered a single mutant mouse strain where the endogenous Ctgf-3' untranslated region (3'UTR) was replaced with a cassette containing two 3'UTR sequences arranged in tandem. The modified Ctgf allele uses a 3'UTR from the mouse FBJ osteosarcoma oncogene (c-Fos) and produces an unstable mRNA, resulting in 60% of normal Ctgf expression (Lo allele). Upon Cre-expression, excision of the c-Fos-3'UTR creates a transcript utilizing the more stable bovine growth hormone (bGH) 3'UTR, resulting in increased Ctgf expression (Hi allele). Using the Ctgf Lo and Hi mutants, and crosses to a Ctgf knockout or Cre-expressing mice, we have generated a series of strains with a 30-fold range of Ctgf expression. Mice with the lowest Ctgf expression, 30% of normal, appear healthy, while a global nine-fold overexpression of Ctgf causes abnormalities, including developmental delay and craniofacial defects, and embryonic death at E10-12. Overexpression of Ctgf by tamoxifen-inducible Cre in the postnatal life, on the other hand, is compatible with life. The Ctgf Lo-Hi mutant mice should prove useful in further understanding the function of CTGF in fibrotic diseases. Additionally, this method can be used for the production of mouse lines with quantitative variations in other genes, particularly with genes that are broadly expressed, have distinct functions in different tissues, or where altered gene expression is not compatible with normal development.
Conflict of interest statement
Figures






References
-
- Thom T, Haase N, Rosamond W, Howard VJ, Rumsfeld J, et al. Heart disease and stroke statistics–2006 update: a report from the American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Circulation. 2006;113:e85–151. - PubMed
-
- González A, López B, Díez J. Myocardial fibrosis in arterial hypertension. European Heart Journal Supplements. 2002;4:D18–D22.
-
- Sun Y, Weber KT. Infarct scar: a dynamic tissue. Cardiovasc Res. 2000;46:250–256. - PubMed
-
- Sun Y, Zhang JQ, Zhang J, Lamparter S. Cardiac remodeling by fibrous tissue after infarction in rats. J Lab Clin Med. 2000;135:316–323. - PubMed
-
- Sun Y, Zhang JQ, Zhang J, Ramires FJ. Angiotensin II, transforming growth factor-beta1 and repair in the infarcted heart. J Mol Cell Cardiol. 1998;30:1559–1569. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

