Human integrin α(3)β(1) regulates TLR2 recognition of lipopeptides from endosomal compartments
- PMID: 20877569
- PMCID: PMC2943923
- DOI: 10.1371/journal.pone.0012871
Human integrin α(3)β(1) regulates TLR2 recognition of lipopeptides from endosomal compartments
Abstract
Background: Toll-like receptor (TLR)-2/TLR1 heterodimers recognize bacterial lipopeptides and initiate the production of inflammatory mediators. Adaptors and co-receptors that mediate this process, as well as the mechanisms by which these adaptors and co-receptors function, are still being discovered.
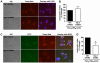
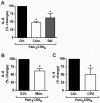
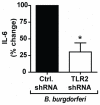
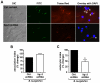
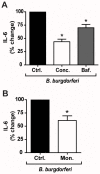
Methodology/principal findings: Using shRNA, blocking antibodies, and fluorescent microscopy, we show that U937 macrophage responses to the TLR2/1 ligand, Pam(3)CSK(4), are dependent upon an integrin, α(3)β(1). The mechanism for integrin α(3)β(1) involvement in TLR2/1 signaling is through its role in endocytosis of lipopeptides. Using inhibitors of endosomal acidification/maturation and physical tethering of the ligand, we show that the endocytosis of Pam(3)CSK(4) is necessary for the complete TLR2/1-mediated pro-inflammatory cytokine response. We also show that TLR2/1 signaling from the endosome results in the induction of different inflammatory mediators than TLR2/1 signaling from the plasma membrane.
Conclusion/significance: Here we identify integrin α(3)β(1) as a novel regulator for the recognition of bacterial lipopeptides. We demonstrate that induction of a specific subset of cytokines is dependent upon integrin α(3)β(1)-mediated endocytosis of the ligand. In addition, we address an ongoing controversy regarding endosomal recognition of bacterial lipopeptides by demonstrating that TLR2/1 signals from within endosomal compartments as well as the plasma membrane, and that downstream responses may differ depending upon receptor localization. We propose that the regulation of endosomal TLR2/1 signaling by integrin α(3)β(1) serves as a mechanism for modulating inflammatory responses.
Conflict of interest statement
Figures

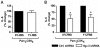

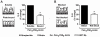


References
-
- Takeuchi O, Akira S. Pattern recognition receptors and inflammation. Cell. 2010;140:805–820. - PubMed
-
- Ferwerda G, Meyer-Wentrup F, Kullberg BJ, Netea MG, Adema GJ. Dectin-1 synergizes with TLR2 and TLR4 for cytokine production in human primary monocytes and macrophages. Cell Microbiol. 2008;10:2058–2066. - PubMed
-
- Schwandner R, Dziarski R, Wesche H, Rothe M, Kirschning CJ. Peptidoglycan- and lipoteichoic acid-induced cell activation is mediated by toll-like receptor 2. J Biol Chem. 1999;274:17406–17409. - PubMed
-
- Yoshimura A, Lien E, Ingalls RR, Tuomanen E, Dziarski R, et al. Cutting edge: recognition of Gram-positive bacterial cell wall components by the innate immune system occurs via Toll-like receptor 2. J Immunol. 1999;163:1–5. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

