Increase of ceftazidime- and fluoroquinolone-resistant Klebsiella pneumoniae and imipenem-resistant Acinetobacter spp. in Korea: analysis of KONSAR study data from 2005 and 2007
- PMID: 20879058
- PMCID: PMC2995968
- DOI: 10.3349/ymj.2010.51.6.901
Increase of ceftazidime- and fluoroquinolone-resistant Klebsiella pneumoniae and imipenem-resistant Acinetobacter spp. in Korea: analysis of KONSAR study data from 2005 and 2007
Abstract
Purpose: Antimicrobial resistance monitoring could be a useful source of information for treating and controlling nosocomial infections. We analyzed antimicrobial resistance data generated by Korean Hospitals and by a commercial laboratory in 2005 and 2007.
Materials and methods: Susceptibility data for 2005 and 2007 were collected from 37 and 41 hospitals, respectively, and from one commercial laboratory. Intermediate susceptibility was not included in the calculation of resistance rates.
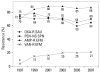
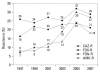
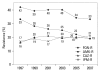
Results: Methicillin-resistant Staphylococcus aureus (MRSA) (64%), third-generation cephalosporin-resistant Klebsiella pneumoniae (29%), fluoroquinolone-resistant Escherichia coli (27%), Pseudomonas aeruginosa (33%), and Acinetobacter spp. (48%), and amikacin-resistant P. aeruginosa (19%) and Acinetobacter spp. (37%) were prevalent in hospitals in 2007. A gradual increase of vancomycin-resistant Enterococcus faecium and imipenem-resistant Acinetobacter spp. was observed. Higher incidences of thirdgeneration cephalosporin-resistant E. coli and K. pneumoniae and imipenemresistant P. aeruginosa were found in the commercial laboratory than in the hospitals.
Conclusion: Methicillin-resistant S. aureus, third-generation cephalosporin- resistant K. pneumoniae, and fluoroquinolone-resistant E. coli, P. aeruginosa and Acinetobacter spp. remain prevalent in Korea, while the incidence of vancomycin-resistant E. faecium and imipenem-resistant Acinetobacter spp. has increased gradually. The higher prevalences of third-generation cephalosporinresistant E. coli and K. pneumoniae, and imipenem-resistant P. aeruginosa in the commercial laboratory are a new concern.
Conflict of interest statement
The authors have no financial conflicts of interest.
Figures

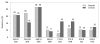
References
-
- Jones RN, Masterton R. Determining the value of antimicrobial surveillance programs. Diagn Microbiol Infect Dis. 2001;41:171–175. - PubMed
-
- Morris AK, Masterton RG. Antibiotic resistance surveillance: action for international studies. J Antimicrob Chemother. 2002;49:7–10. - PubMed
-
- Cornaglia G, Hryniewicz W, Jarlier V, Kahlmeter G, Mittermayer H, Stratchounski L, et al. European recommendations for antimicrobial resistance surveillance. Clin Microbiol Infect. 2004;10:349–383. - PubMed
-
- Llata E, Gaynes RP, Fridkin S. Measuring the scope and magnitude of hospital-associated infection in the United States: the value of prevalence surveys. Clin Infect Dis. 2009;48:1434–1440. - PubMed
-
- Chong Y, Lee K, Park YJ, Jeon DS, Lee MH, Kim MY, et al. Korean Nationwide Surveillance of Antimicrobial Resistance of bacteria in 1997. Yonsei Med J. 1998;39:569–577. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

