Development of stable phosphohistidine analogues
- PMID: 20879710
- PMCID: PMC3189792
- DOI: 10.1021/ja104393t
Development of stable phosphohistidine analogues
Abstract
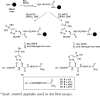
Protein phosphorylation is one of the most common and extensively studied posttranslational modifications (PTMs). Compared to the O-phosphorylation of Ser, Thr, and Tyr residues, our understanding of histidine phosphorylation is relatively limited, particularly in higher eukaryotes, due to technical difficulties stemming from the intrinsic instability and isomerism of phosphohistidine (pHis). We report the design and synthesis of stable and nonisomerizable pHis analogues. These pHis analogues were successfully utilized in solid-phase peptide synthesis and semi-synthesis of histone H4. Significantly, the first antibody that specifically recognizes pHis was obtained using the synthetic peptide as the immunogen.
Figures




References
-
- Walsh CT. Posttranslational Modification of Proteins: Expanding Nature's Inventory. Roberts and Company Publishers; Greenwood Village: 2006. p. 490.
-
- Druker BJ. Oncologist. 2004;9:357–360. - PubMed
-
- Cohen P. Nat. Rev. Drug Discovery. 2002;1:309–315. - PubMed
-
- Khorchid A, Ikura M. Int. J. Biochem. Cell Biol. 2006;38:307–312. - PubMed
-
- Zetterqvist O, Engstrom L. Biochim. Biophys. Acta. 1966;113:520–530. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

