Maternal care and DNA methylation of a glutamic acid decarboxylase 1 promoter in rat hippocampus
- PMID: 20881131
- PMCID: PMC6633524
- DOI: 10.1523/JNEUROSCI.1039-10.2010
Maternal care and DNA methylation of a glutamic acid decarboxylase 1 promoter in rat hippocampus
Abstract
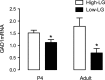
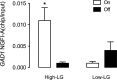
Parenting and the early environment influence the risk for various psychopathologies. Studies in the rat suggest that variations in maternal care stably influence DNA methylation, gene expression, and neural function in the offspring. Maternal care affects neural development, including the GABAergic system, the function of which is linked to the pathophysiology of diseases including schizophrenia and depression. Postmortem studies of human schizophrenic brains have revealed decreased forebrain expression of glutamic acid decarboxylase 1 (GAD1) accompanied by increased methylation of a GAD1 promoter. We examined whether maternal care affects GAD1 promoter methylation in the hippocampus of adult male offspring of high and low pup licking/grooming (high-LG and low-LG) mothers. Compared with the offspring of low-LG mothers, those reared by high-LG dams showed enhanced hippocampal GAD1 mRNA expression, decreased cytosine methylation, and increased histone 3-lysine 9 acetylation (H3K9ac) of the GAD1 promoter. DNA methyltransferase 1 expression was significantly higher in the offspring of low- compared with high-LG mothers. Pup LG increases hippocampal serotonin (5-HT) and nerve growth factor-inducible factor A (NGFI-A) expression. Chromatin immunoprecipitation assays revealed enhanced NGFI-A association with and H3K9ac of the GAD1 promoter in the hippocampus of high-LG pups after a nursing bout. Treatment of hippocampal neuronal cultures with either 5-HT or an NGFI-A expression plasmid significantly increased GAD1 mRNA levels. The effect of 5-HT was blocked by a short interfering RNA targeting NGFI-A. These results suggest that maternal care influences the development of the GABA system by altering GAD1 promoter methylation levels through the maternally induced activation of NGFI-A and its association with the GAD1 promoter.
Figures



Similar articles
-
The transcription factor nerve growth factor-inducible protein a mediates epigenetic programming: altering epigenetic marks by immediate-early genes.J Neurosci. 2007 Feb 14;27(7):1756-68. doi: 10.1523/JNEUROSCI.4164-06.2007. J Neurosci. 2007. PMID: 17301183 Free PMC article.
-
Maternal licking regulates hippocampal glucocorticoid receptor transcription through a thyroid hormone-serotonin-NGFI-A signalling cascade.Philos Trans R Soc Lond B Biol Sci. 2012 Sep 5;367(1601):2495-510. doi: 10.1098/rstb.2012.0223. Philos Trans R Soc Lond B Biol Sci. 2012. PMID: 22826348 Free PMC article.
-
Variations in postnatal maternal care and the epigenetic regulation of metabotropic glutamate receptor 1 expression and hippocampal function in the rat.Proc Natl Acad Sci U S A. 2012 Oct 16;109 Suppl 2(Suppl 2):17200-7. doi: 10.1073/pnas.1204599109. Epub 2012 Oct 8. Proc Natl Acad Sci U S A. 2012. PMID: 23045678 Free PMC article.
-
Maternal programming of steroid receptor expression and phenotype through DNA methylation in the rat.Front Neuroendocrinol. 2005 Oct-Dec;26(3-4):139-62. doi: 10.1016/j.yfrne.2005.10.002. Epub 2005 Nov 21. Front Neuroendocrinol. 2005. PMID: 16303171 Review.
-
Epigenetic mechanisms for the early environmental regulation of hippocampal glucocorticoid receptor gene expression in rodents and humans.Neuropsychopharmacology. 2013 Jan;38(1):111-23. doi: 10.1038/npp.2012.149. Epub 2012 Sep 12. Neuropsychopharmacology. 2013. PMID: 22968814 Free PMC article. Review.
Cited by
-
A steep learning curve: decoding epigenetic influence on behavior and mental health.Environ Health Perspect. 2012 Oct;120(10):a396-401. doi: 10.1289/ehp.120-a396. Environ Health Perspect. 2012. PMID: 23026389 Free PMC article. No abstract available.
-
Epigenetics and Depression: An Update.Psychiatry Investig. 2019 Sep;16(9):654-661. doi: 10.30773/pi.2019.07.17.2. Epub 2019 Aug 29. Psychiatry Investig. 2019. PMID: 31455063 Free PMC article.
-
Prevention of age-related changes in hippocampal levels of 5-methylcytidine by caloric restriction.Neurobiol Aging. 2012 Aug;33(8):1672-81. doi: 10.1016/j.neurobiolaging.2011.06.003. Epub 2011 Jul 18. Neurobiol Aging. 2012. PMID: 21764481 Free PMC article.
-
Epigenetic medicine and fetal alcohol spectrum disorders.Epigenomics. 2013 Feb;5(1):73-86. doi: 10.2217/epi.12.80. Epigenomics. 2013. PMID: 23414322 Free PMC article. Review.
-
Global and gene-specific DNA methylation alterations in the adolescent amygdala and hippocampus in an animal model of caregiver maltreatment.Behav Brain Res. 2016 Feb 1;298(Pt A):55-61. doi: 10.1016/j.bbr.2015.05.028. Epub 2015 May 28. Behav Brain Res. 2016. PMID: 26027495 Free PMC article.
References
-
- Akbarian S, Kim JJ, Potkin SG, Hagman JO, Tafazzoli A, Bunney WE, Jr, Jones EG. Gene expression for glutamic acid decarboxylase is reduced without loss of neurons in prefrontal cortex of schizophrenics. Arch Gen Psychiatry. 1995;52:258–266. - PubMed
-
- Benes FM. The role of stress and dopamine-GABA interactions in the vulnerability for schizophrenia. J Psychiatr Res. 1997;31:257–275. - PubMed
-
- Bird AP. CpG-rich islands and the function of DNA methylation. Nature. 1986;321:209–213. - PubMed
-
- Brandeis M, Frank D, Keshet I, Siegfried Z, Mendelsohn M, Nemes A, Temper V, Razin A, Cedar H. Spl elements protect a CpG island from de novo methylation. Nature. 1994;371:435–438. - PubMed
-
- Buryanov YI, Shevchuk TV. DNA methyltransferases and structural-functional specificity of eukaryotic DNA modification. Biochemistry (Mosc) 2005;70:730–742. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
