Tannerella forsythia infection-induced calvarial bone and soft tissue transcriptional profiles
- PMID: 20883221
- PMCID: PMC3407412
- DOI: 10.1111/j.2041-1014.2010.00583.x
Tannerella forsythia infection-induced calvarial bone and soft tissue transcriptional profiles
Abstract
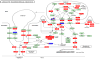
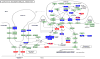
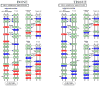
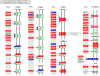
Tannerella forsythia is associated with subgingival biofilms in adult periodontitis, although the molecular mechanisms contributing to chronic inflammation and loss of periodontal bone remain unclear. We examined changes in the host transcriptional profiles during a T. forsythia infection using a murine calvarial model of inflammation and bone resorption. Tannerella forsythia was injected into the subcutaneous soft tissue over calvariae of BALB/c mice for 3 days, after which the soft tissues and calvarial bones were excised. RNA was isolated and Murine GeneChip (Affymetrix, Santa Clara, CA) array analysis of transcript profiles showed that 3226 genes were differentially expressed in the infected soft tissues (P < 0.05) and 2586 genes were differentially transcribed in calvarial bones after infection. Quantitative real-time reverse transcription-polymerase chain reaction analysis of transcription levels of selected genes corresponded well with the microarray results. Biological pathways significantly impacted by T. forsythia infection in calvarial bone and soft tissue included leukocyte transendothelial migration, cell adhesion molecules (immune system), extracellular matrix-receptor interaction, adherens junction, and antigen processing and presentation. Histologic examination revealed intense inflammation and increased osteoclasts in calvariae compared with controls. In conclusion, localized T. forsythia infection differentially induces transcription of a broad array of host genes, and the profiles differ between inflamed soft tissues and calvarial bone.
© 2010 John Wiley & Sons A/S.
Conflict of interest statement
The authors have no financial conflict of interest.
Figures



Similar articles
-
Polymicrobial periodontal pathogen transcriptomes in calvarial bone and soft tissue.Mol Oral Microbiol. 2011 Oct;26(5):303-20. doi: 10.1111/j.2041-1014.2011.00619.x. Epub 2011 Jul 19. Mol Oral Microbiol. 2011. PMID: 21896157 Free PMC article.
-
Molecular characterization of Treponema denticola infection-induced bone and soft tissue transcriptional profiles.Mol Oral Microbiol. 2010 Aug;25(4):260-74. doi: 10.1111/j.2041-1014.2010.00575.x. Mol Oral Microbiol. 2010. PMID: 20618700 Free PMC article.
-
Porphyromonas gingivalis infection-induced tissue and bone transcriptional profiles.Mol Oral Microbiol. 2010 Feb;25(1):61-74. doi: 10.1111/j.2041-1014.2009.00555.x. Mol Oral Microbiol. 2010. PMID: 20331794 Free PMC article.
-
Tannerella forsythia GroEL induces inflammatory bone resorption and synergizes with interleukin-17.Mol Oral Microbiol. 2017 Aug;32(4):301-313. doi: 10.1111/omi.12172. Epub 2016 Sep 13. Mol Oral Microbiol. 2017. PMID: 27484636
-
TLR2 signaling and Th2 responses drive Tannerella forsythia-induced periodontal bone loss.J Immunol. 2011 Jul 1;187(1):501-9. doi: 10.4049/jimmunol.1100683. Epub 2011 Jun 1. J Immunol. 2011. PMID: 21632710 Free PMC article.
Cited by
-
Polymicrobial Oral Infection with Four Periodontal Bacteria Orchestrates a Distinct Inflammatory Response and Atherosclerosis in ApoE null Mice.PLoS One. 2015 Nov 30;10(11):e0143291. doi: 10.1371/journal.pone.0143291. eCollection 2015. PLoS One. 2015. PMID: 26619277 Free PMC article.
-
Polymicrobial periodontal pathogen transcriptomes in calvarial bone and soft tissue.Mol Oral Microbiol. 2011 Oct;26(5):303-20. doi: 10.1111/j.2041-1014.2011.00619.x. Epub 2011 Jul 19. Mol Oral Microbiol. 2011. PMID: 21896157 Free PMC article.
-
Exploring the Oral Microbiome: Understanding its Impact on the Development of Oral Squamous Cell Carcinoma.Curr Microbiol. 2025 Aug 16;82(10):458. doi: 10.1007/s00284-025-04377-w. Curr Microbiol. 2025. PMID: 40819017 Review.
-
Animal models to study host-bacteria interactions involved in periodontitis.Front Oral Biol. 2012;15:117-32. doi: 10.1159/000329675. Epub 2011 Nov 11. Front Oral Biol. 2012. PMID: 22142960 Free PMC article. Review.
References
-
- Boyce BF, Aufdemorte TB, Garrett IR, Yates AJ, Mundy GR. Effects of interleukin-1 on bone turnover in normal mice. Endocrinology. 1989;125:1142–1150. - PubMed
-
- Cohen P, Bouaboula M, Bellis M, et al. Monitoring cellular responses to Listeria monocytogenes with oligonucleotide arrays. J Biol Chem. 2000;275:11181–11190. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

