Paracrine stimulation of endothelial cell motility and angiogenesis by platelet-derived deoxyribose-1-phosphate
- PMID: 20884872
- PMCID: PMC7528120
- DOI: 10.1161/ATVBAHA.110.215855
Paracrine stimulation of endothelial cell motility and angiogenesis by platelet-derived deoxyribose-1-phosphate
Abstract
Objective: Micromolar concentrations of the proangiogenic metabolite deoxyribose-1-phosphate (dRP) were detected in platelet supernatants by mass spectrometry. In this study, we assessed whether the release of dRP by platelets stimulates endothelial cell migration and angiogenesis.
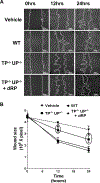
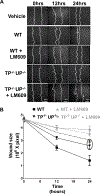
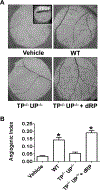
Methods and results: Protein-free supernatants from thrombin-stimulated platelets increased human umbilical vein endothelial cell migratory activity in transmigration and monolayer repair assays. This phenomenon was ablated by genetic silencing of dRP-generating uridine phosphorylase (UP) and thymidine phosphorylase (TP) or pharmacological inhibition of UP and restored by exogenous dRP. The stimulation of endothelial cell migration by platelet-derived dRP correlated with upregulation of integrin β(3), which was induced in a reactive oxygen species-dependent manner, and was mediated by the activity of the integrin heterodimer α(v)β(3). The physiological relevance of dRP release by platelets was confirmed in a chick chorioallantoic membrane assay, where the presence of this metabolite in platelet supernatants strongly induced capillary formation.
Conclusions: Platelet-derived dRP stimulates endothelial cell migration by upregulating integrin β(3) in a reactive oxygen species-dependent manner. As demonstrated by our in vivo experiments, this novel paracrine regulatory pathway is likely to play an important role in the stimulation of angiogenesis by platelets.
Figures



References
-
- Brill A, Elinav H, Varon D. Differential role of platelet granular mediators in angiogenesis. Cardiovasc Res. 2004;63:226–235. - PubMed
-
- Brill A, Dashevsky O, Rivo J, Gozal Y, Varon D. Platelet-derived microparticles induce angiogenesis and stimulate post-ischemic revascularization. Cardiovasc Res. 2005;67:30–38. - PubMed
-
- Rhee JS, Black M, Schubert U, Fischer S, Morgenstern E, Hammes HP, Preissner KT. The functional role of blood platelet components in angiogenesis. Thromb Haemost. 2004;92:394–402. - PubMed
-
- Nurden AT, Nurden P, Sanchez M, Andia I, Anitua E. Platelets and wound healing. Front Biosci. 2008;13:3532–3548. - PubMed
-
- Langer HF, Gawaz M. Platelets in regenerative medicine. Basic Res Cardiol. 2008;103:299–307. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

