Genomic analysis of carbon monoxide utilization and butanol production by Clostridium carboxidivorans strain P7
- PMID: 20885952
- PMCID: PMC2946384
- DOI: 10.1371/journal.pone.0013033
Genomic analysis of carbon monoxide utilization and butanol production by Clostridium carboxidivorans strain P7
Abstract
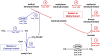
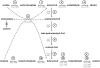
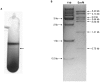
Increasing demand for the production of renewable fuels has recently generated a particular interest in microbial production of butanol. Anaerobic bacteria, such as Clostridium spp., can naturally convert carbohydrates into a variety of primary products, including alcohols like butanol. The genetics of microorganisms like Clostridium acetobutylicum have been well studied and their solvent-producing metabolic pathways characterized. In contrast, less is known about the genetics of Clostridium spp. capable of converting syngas or its individual components into solvents. In this study, the type of strain of a new solventogenic Clostridium species, C. carboxidivorans, was genetically characterized by genome sequencing. C. carboxidivorans strain P7(T) possessed a complete Wood-Ljungdahl pathway gene cluster, involving CO and CO(2) fixation and conversion to acetyl-CoA. Moreover, with the exception of an acetone production pathway, all the genetic determinants of canonical ABE metabolic pathways for acetate, butyrate, ethanol and butanol production were present in the P7(T) chromosome. The functionality of these pathways was also confirmed by growth of P7(T) on CO and production of CO(2) as well as volatile fatty acids (acetate and butyrate) and solvents (ethanol and butanol). P7(T) was also found to harbour a 19 Kbp plasmid, which did not include essential or butanol production related genes. This study has generated in depth knowledge of the P7(T) genome, which will be helpful in developing metabolic engineering strategies to improve C. carboxidivorans's natural capacity to produce potential biofuels from syngas.
Conflict of interest statement
Figures




References
-
- Schwarz WH, Gapes JR. Butanol – rediscovering a renewable fuel. BioWorld Europe. 2006;01-2006:16–19.
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

