Septins from the phytopathogenic fungus Ustilago maydis are required for proper morphogenesis but dispensable for virulence
- PMID: 20885997
- PMCID: PMC2946335
- DOI: 10.1371/journal.pone.0012933
Septins from the phytopathogenic fungus Ustilago maydis are required for proper morphogenesis but dispensable for virulence
Abstract
Background: Septins are a highly conserved family of GTP-binding proteins involved in multiple cellular functions, including cell division and morphogenesis. Studies of septins in fungal cells underpin a clear correlation between septin-based structures and fungal morphology, providing clues to understand the molecular frame behind the varied morphologies found in fungal world.
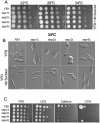
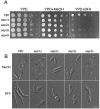
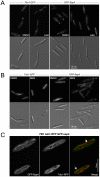
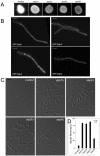
Methodology/principal findings: Ustilago maydis genome has the ability to encode four septins. Here, using loss-of-function as well as GFP-tagged alleles of these septin genes, we investigated the roles of septins in the morphogenesis of this basidiomycete fungus. We described that septins in U. maydis could assemble into at least three different structures coexisting in the same cell: bud neck collars, band-like structures at the growing tip, and long septin fibers that run from pole to pole near the cell cortex. We also found that in the absence of septins, U. maydis cells lost their elongated shape, became wider at the central region and ended up losing their polarity, pointing to an important role of septins in the morphogenesis of this fungus. These morphological defects were alleviated in the presence of an osmotic stabilizer suggesting that absence of septins affected the proper formation of the cell wall, which was coherent with a higher sensitivity of septin defective cells to drugs that affect cell wall construction as well as exocytosis. As U. maydis is a phytopathogen, we analyzed the role of septins in virulence and found that in spite of the described morphological defects, septin mutants were virulent in corn plants.
Conclusions/significance: Our results indicated a major role of septins in morphogenesis in U. maydis. However, in contrast to studies in other fungal pathogens, in which septins were reported to be necessary during the infection process, we found a minor role of septins during corn infection by U. maydis.
Conflict of interest statement
Figures






Similar articles
-
Spa2 is required for morphogenesis but it is dispensable for pathogenicity in the phytopathogenic fungus Ustilago maydis.Fungal Genet Biol. 2008 Sep;45(9):1315-27. doi: 10.1016/j.fgb.2008.06.010. Epub 2008 Jul 11. Fungal Genet Biol. 2008. PMID: 18674629
-
Sustained cell polarity and virulence in the phytopathogenic fungus Ustilago maydis depends on an essential cyclin-dependent kinase from the Cdk5/Pho85 family.J Cell Sci. 2007 May 1;120(Pt 9):1584-95. doi: 10.1242/jcs.005314. Epub 2007 Apr 3. J Cell Sci. 2007. PMID: 17405809
-
The core effector Cce1 is required for early infection of maize by Ustilago maydis.Mol Plant Pathol. 2018 Oct;19(10):2277-2287. doi: 10.1111/mpp.12698. Epub 2018 Aug 16. Mol Plant Pathol. 2018. PMID: 29745456 Free PMC article.
-
The machinery for cell polarity, cell morphogenesis, and the cytoskeleton in the Basidiomycete fungus Ustilago maydis-a survey of the genome sequence.Fungal Genet Biol. 2008 Aug;45 Suppl 1(Suppl 1):S3-S14. doi: 10.1016/j.fgb.2008.05.012. Epub 2008 Jun 7. Fungal Genet Biol. 2008. PMID: 18582586 Free PMC article. Review.
-
Protein glycosylation in the phytopathogen Ustilago maydis: From core oligosaccharide synthesis to the ER glycoprotein quality control system, a genomic analysis.Fungal Genet Biol. 2010 Sep;47(9):727-35. doi: 10.1016/j.fgb.2010.06.004. Epub 2010 Jun 8. Fungal Genet Biol. 2010. PMID: 20554055 Review.
Cited by
-
Dephosphorylation of the Core Septin, AspB, in a Protein Phosphatase 2A-Dependent Manner Impacts Its Localization and Function in the Fungal Pathogen Aspergillus fumigatus.Front Microbiol. 2016 Jun 22;7:997. doi: 10.3389/fmicb.2016.00997. eCollection 2016. Front Microbiol. 2016. PMID: 27446037 Free PMC article.
-
Appressorium formation in the corn smut fungus Ustilago maydis requires a G2 cell cycle arrest.Plant Signal Behav. 2015;10(4):e1001227. doi: 10.1080/15592324.2014.1001227. Plant Signal Behav. 2015. PMID: 25876077 Free PMC article.
-
Endosomal transport of septin mRNA and protein indicates local translation on endosomes and is required for correct septin filamentation.EMBO Rep. 2014 Jan;15(1):94-102. doi: 10.1002/embr.201338037. Epub 2013 Dec 15. EMBO Rep. 2014. PMID: 24355572 Free PMC article.
-
Control of septin filament flexibility and bundling by subunit composition and nucleotide interactions.Mol Biol Cell. 2018 Mar 15;29(6):702-712. doi: 10.1091/mbc.E17-10-0608. Epub 2018 Jan 10. Mol Biol Cell. 2018. PMID: 29321251 Free PMC article.
-
Fungal pathogens are platforms for discovering novel and conserved septin properties.Curr Opin Microbiol. 2014 Aug;20:42-8. doi: 10.1016/j.mib.2014.04.004. Epub 2014 May 28. Curr Opin Microbiol. 2014. PMID: 24879478 Free PMC article. Review.
References
-
- Hartwell LH. Genetic control of the cell division cycle in yeast. IV. Genes controlling bud emergence and cytokinesis. Exp Cell Res. 1971;69:265–276. - PubMed
-
- Caudron F, Barral Y. Septins and the lateral compartmentalization of eukaryotic membranes. Dev Cell. 2009;16:493–506. - PubMed
-
- Faty M, Fink M, Barral Y. Septins: a ring to part mother and daughter. Curr Genet. 2002;41:123–131. - PubMed
-
- Gladfelter AS. Control of filamentous fungal cell shape by septins and formins. Nat Rev Microbiol. 2006;4:223–229. - PubMed
-
- Kinoshita M. Diversity of septin scaffolds. Curr Opin Cell Biol. 2006;18:54–60. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

