Fourier-Bessel reconstruction of helical assemblies
- PMID: 20888960
- PMCID: PMC3128834
- DOI: 10.1016/S0076-6879(10)82005-1
Fourier-Bessel reconstruction of helical assemblies
Abstract
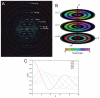
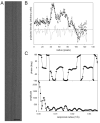
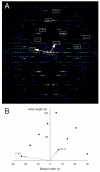
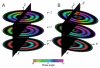
Helical symmetry is commonly used for building macromolecular assemblies. Helical symmetry is naturally present in viruses and cytoskeletal filaments and also occurs during crystallization of isolated proteins, such as Ca-ATPase and the nicotinic acetyl choline receptor. Structure determination of helical assemblies by electron microscopy has a long history dating back to the original work on three-dimensional (3D) reconstruction. A helix offers distinct advantages for structure determination. Not only can one improve resolution by averaging across the constituent subunits, but each helical assembly provides multiple views of these subunits and thus provides a complete 3D data set. This review focuses on Fourier methods of helical reconstruction, covering the theoretical background, a step-by-step guide to the process, and a practical example based on previous work with Ca-ATPase. Given recent results from helical reconstructions at atomic resolution and the development of graphical user interfaces to aid in the process, these methods are likely to continue to make an important contribution to the field of structural biology.
Copyright © 2010 Elsevier Inc. All rights reserved.
Figures






Similar articles
-
Python-based Helix Indexer: A graphical user interface program for finding symmetry of helical assembly through Fourier-Bessel indexing of electron microscopic data.Protein Sci. 2022 Jan;31(1):107-117. doi: 10.1002/pro.4186. Epub 2021 Sep 22. Protein Sci. 2022. PMID: 34529294 Free PMC article.
-
Cryo-EM Structure Determination Using Segmented Helical Image Reconstruction.Methods Enzymol. 2016;579:307-28. doi: 10.1016/bs.mie.2016.05.034. Epub 2016 Jun 28. Methods Enzymol. 2016. PMID: 27572732 Review.
-
Single-particle reconstruction from EM images of helical filaments.Curr Opin Struct Biol. 2007 Oct;17(5):556-61. doi: 10.1016/j.sbi.2007.07.006. Epub 2007 Sep 11. Curr Opin Struct Biol. 2007. PMID: 17851070 Free PMC article. Review.
-
Reconstruction of helical filaments and tubes.Methods Enzymol. 2010;482:167-83. doi: 10.1016/S0076-6879(10)82006-3. Methods Enzymol. 2010. PMID: 20888961 Free PMC article.
-
SPRING - an image processing package for single-particle based helical reconstruction from electron cryomicrographs.J Struct Biol. 2014 Jan;185(1):15-26. doi: 10.1016/j.jsb.2013.11.003. Epub 2013 Nov 21. J Struct Biol. 2014. PMID: 24269218
Cited by
-
Calcium sensitive ring-like oligomers formed by synaptotagmin.Proc Natl Acad Sci U S A. 2014 Sep 23;111(38):13966-71. doi: 10.1073/pnas.1415849111. Epub 2014 Sep 8. Proc Natl Acad Sci U S A. 2014. PMID: 25201968 Free PMC article.
-
Outcome of the first electron microscopy validation task force meeting.Structure. 2012 Feb 8;20(2):205-14. doi: 10.1016/j.str.2011.12.014. Structure. 2012. PMID: 22325770 Free PMC article.
-
Real-space processing of helical filaments in SPARX.J Struct Biol. 2012 Feb;177(2):302-13. doi: 10.1016/j.jsb.2011.12.020. Epub 2012 Jan 11. J Struct Biol. 2012. PMID: 22248449 Free PMC article.
-
Deducing the symmetry of helical assemblies: Applications to membrane proteins.J Struct Biol. 2016 Aug;195(2):167-178. doi: 10.1016/j.jsb.2016.05.011. Epub 2016 May 30. J Struct Biol. 2016. PMID: 27255388 Free PMC article.
-
Inward-facing conformation of the zinc transporter YiiP revealed by cryoelectron microscopy.Proc Natl Acad Sci U S A. 2013 Feb 5;110(6):2140-5. doi: 10.1073/pnas.1215455110. Epub 2013 Jan 22. Proc Natl Acad Sci U S A. 2013. PMID: 23341604 Free PMC article.
References
-
- Beroukhim R, Unwin N. Distortion correction of tubular crystals: Improvements in the acetylcholine receptor structure. Ultramicroscopy. 1997;70:57–81. - PubMed
-
- Castellani L, Hardwicke PM, Vibert P. Dimer ribbons in the three-dimensional structure of sarcoplasmic reticulum. J. Mol. Biol. 1985;185:579–594. - PubMed
-
- Chen YJ, Zhang P, Egelman EH, Hinshaw JE. The stalk region of dynamin drives the constriction of dynamin tubes. Nat Struct Mol Biol. 2004;11:574–5. - PubMed
-
- Cochran W, Crick FHC, Vand V. The Structure of Synthetic Polypeptides. I. The Transform of Atoms on a Helix. Acta Cryst. 1952;5:581–586.
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

