Alkyltransferase-like protein (eATL) prevents mismatch repair-mediated toxicity induced by O6-alkylguanine adducts in Escherichia coli
- PMID: 20921378
- PMCID: PMC2964255
- DOI: 10.1073/pnas.1008635107
Alkyltransferase-like protein (eATL) prevents mismatch repair-mediated toxicity induced by O6-alkylguanine adducts in Escherichia coli
Abstract
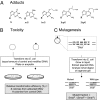
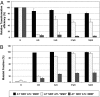
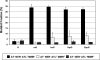
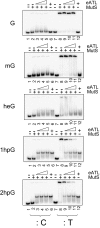
O(6)-alkylG adducts are highly mutagenic due to their capacity to efficiently form O(6)-alkylG:T mispairs during replication, thus triggering G→A transitions. Mutagenesis is largely prevented by repair strategies such as reversal by alkyltransferases or excision by nucleotide excision repair (NER). Moreover, methyl-directed mismatch repair (MMR) is known to trigger sensitivity to methylating agents via a mechanism that involves recognition by MutS of the O(6)-mG:T replication intermediates. We wanted to investigate the mechanism by which MMR controls the genotoxicity of environmentally relevant O(6)-alkylG adducts formed by ethylene oxide and propylene oxide. Recently, the alkyltransferase-like gene ybaZ (eATL) was shown to enhance repair of these slightly larger O(6)-alkylG adducts by NER. We analyzed the toxicity and mutagenesis induced by these O(6)-alkylG adducts using single-adducted plasmid probes. We show that the eATL gene product prevents MMR-mediated attack of the O(6)-alkylG:T replication intermediate for the larger alkyl groups but not for methyl. In vivo data are compatible with the occurrence of repeated cycles of MMR attack of the O(6)-alkylG:T intermediate. In addition, in vitro, the eATL protein efficiently prevents binding of MutS to the O(6)-alkylG:T mispairs formed by the larger alkyl groups but not by methyl. In conclusion, eATL not only enhances the efficiency of repair of these larger adducts by NER, it also shields these adducts from MMR-mediated toxicity.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
The bacterial alkyltransferase-like (eATL) protein protects mammalian cells against methylating agent-induced toxicity.DNA Repair (Amst). 2015 Apr;28:14-20. doi: 10.1016/j.dnarep.2015.01.009. Epub 2015 Jan 31. DNA Repair (Amst). 2015. PMID: 25703834
-
The alkyltransferase-like ybaZ gene product enhances nucleotide excision repair of O(6)-alkylguanine adducts in E. coli.DNA Repair (Amst). 2009 Jun 4;8(6):697-703. doi: 10.1016/j.dnarep.2009.01.022. Epub 2009 Mar 9. DNA Repair (Amst). 2009. PMID: 19269902
-
E. coli mismatch repair enhances AT-to-GC mutagenesis caused by alkylating agents.Mutat Res Genet Toxicol Environ Mutagen. 2017 Mar;815:22-27. doi: 10.1016/j.mrgentox.2017.02.001. Epub 2017 Feb 15. Mutat Res Genet Toxicol Environ Mutagen. 2017. PMID: 28283089
-
Alkylation damage in DNA and RNA--repair mechanisms and medical significance.DNA Repair (Amst). 2004 Nov 2;3(11):1389-407. doi: 10.1016/j.dnarep.2004.05.004. DNA Repair (Amst). 2004. PMID: 15380096 Review.
-
Repair of O(6)-alkylguanine by alkyltransferases.Mutat Res. 2000 Apr;462(2-3):83-100. doi: 10.1016/s1383-5742(00)00017-x. Mutat Res. 2000. PMID: 10767620 Review.
Cited by
-
Alkyltransferase-like protein (Atl1) distinguishes alkylated guanines for DNA repair using cation-π interactions.Proc Natl Acad Sci U S A. 2012 Nov 13;109(46):18755-60. doi: 10.1073/pnas.1209451109. Epub 2012 Oct 29. Proc Natl Acad Sci U S A. 2012. PMID: 23112169 Free PMC article.
-
Alkyltransferase-like protein clusters scan DNA rapidly over long distances and recruit NER to alkyl-DNA lesions.Proc Natl Acad Sci U S A. 2020 Apr 28;117(17):9318-9328. doi: 10.1073/pnas.1916860117. Epub 2020 Apr 9. Proc Natl Acad Sci U S A. 2020. PMID: 32273391 Free PMC article.
-
Crosstalk between repair pathways elicits double-strand breaks in alkylated DNA and implications for the action of temozolomide.Elife. 2021 Jul 8;10:e69544. doi: 10.7554/eLife.69544. Elife. 2021. PMID: 34236314 Free PMC article.
-
The aminoglycoside antibiotic kanamycin damages DNA bases in Escherichia coli: caffeine potentiates the DNA-damaging effects of kanamycin while suppressing cell killing by ciprofloxacin in Escherichia coli and Bacillus anthracis.Antimicrob Agents Chemother. 2012 Jun;56(6):3216-23. doi: 10.1128/AAC.00066-12. Epub 2012 Mar 5. Antimicrob Agents Chemother. 2012. PMID: 22391551 Free PMC article.
-
Understanding the importance of low-molecular weight (ethylene oxide- and propylene oxide-induced) DNA adducts and mutations in risk assessment: Insights from 15 years of research and collaborative discussions.Environ Mol Mutagen. 2019 Mar;60(2):100-121. doi: 10.1002/em.22248. Epub 2018 Dec 10. Environ Mol Mutagen. 2019. PMID: 30536466 Free PMC article. Review.
References
-
- Delaney JC, Essigmann JM. Effect of sequence context on O(6)-methylguanine repair and replication in vivo. Biochemistry. 2001;40:14968–14975. - PubMed
-
- Hill-Perkins M, Jones MD, Karran P. Site-specific mutagenesis in vivo by single methylated or deaminated purine bases. Mutat Res. 1986;162:153–163. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

