Inhibition of brain tumor growth by intravenous poly (β-L-malic acid) nanobioconjugate with pH-dependent drug release [corrected]
- PMID: 20921419
- PMCID: PMC2964197
- DOI: 10.1073/pnas.1003919107
Inhibition of brain tumor growth by intravenous poly (β-L-malic acid) nanobioconjugate with pH-dependent drug release [corrected]
Erratum in
- Proc Natl Acad Sci U S A. 2010 Nov 9;107(45):19603
Abstract
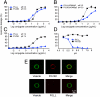
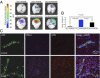
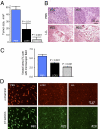
Effective treatment of brain neurological disorders such as Alzheimer's disease, multiple sclerosis, or tumors should be possible with drug delivery through blood-brain barrier (BBB) or blood-brain tumor barrier (BTB) and targeting specific types of brain cells with drug release into the cell cytoplasm. A polymeric nanobioconjugate drug based on biodegradable, nontoxic, and nonimmunogenic polymalic acid as a universal delivery nanoplatform was used for design and synthesis of nanomedicine drug for i.v. treatment of brain tumors. The polymeric drug passes through the BTB and tumor cell membrane using tandem monoclonal antibodies targeting the BTB and tumor cells. The next step for polymeric drug action was inhibition of tumor angiogenesis by specifically blocking the synthesis of a tumor neovascular trimer protein, laminin-411, by attached antisense oligonucleotides (AONs). The AONs were released into the target cell cytoplasm via pH-activated trileucine, an endosomal escape moiety. Drug delivery to the brain tumor and the release mechanism were both studied for this nanobiopolymer. Introduction of a trileucine endosome escape unit resulted in significantly increased AON delivery to tumor cells, inhibition of laminin-411 synthesis in vitro and in vivo, specific accumulation in brain tumors, and suppression of intracranial glioma growth compared with pH-independent leucine ester. The availability of a systemically active polymeric drug delivery system that passes through the BTB, targets tumor cells, and inhibits glioma growth gives hope for a successful strategy of glioma treatment. This delivery system with drug release into the brain-specific cell type could be useful for treatment of various brain pathologies.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Comment in
-
Drug delivery: Nanobioconjugate shrinks brain tumours.Nat Rev Drug Discov. 2010 Dec;9(12):917. doi: 10.1038/nrd3332. Epub 2010 Nov 19. Nat Rev Drug Discov. 2010. PMID: 21088679 No abstract available.
References
-
- Brandsma D, van den Bent MJ. Molecular targeted therapies and chemotherapy in malignant gliomas. Curr Opin Oncol. 2007;19:598–605. - PubMed
-
- Folkman J. Angiogenesis: An organizing principle for drug discovery? Nat Rev Drug Discov. 2007;6:273–286. - PubMed
-
- Ljubimova JY, et al. Overexpression of alpha4 chain-containing laminins in human glial tumors identified by gene microarray analysis. Cancer Res. 2001;61:5601–5610. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

