Phase II selection design trial of concurrent chemotherapy and cetuximab versus chemotherapy followed by cetuximab in advanced-stage non-small-cell lung cancer: Southwest Oncology Group study S0342
- PMID: 20921467
- PMCID: PMC3020704
- DOI: 10.1200/JCO.2009.27.9356
Phase II selection design trial of concurrent chemotherapy and cetuximab versus chemotherapy followed by cetuximab in advanced-stage non-small-cell lung cancer: Southwest Oncology Group study S0342
Abstract
Purpose: Randomized clinical trials failed to show a survival benefit for epidermal growth factor receptor (EGFR) tyrosine kinase inhibitors plus concurrent chemotherapy in patients with metastatic non-small-cell lung cancer (NSCLC), with preclinical data suggesting potential negative interactions. In contrast, pilot trials of the EGFR-targeted antibody, cetuximab, plus chemotherapy suggested enhanced antitumor activity. This randomized phase II trial was designed to select a cetuximab plus chemotherapy regimen for phase III evaluation.
Patients and methods: Treatment-naive patients with advanced-stage NSCLC were randomly assigned to receive paclitaxel (225 mg/m(2)) and carboplatin (area under the curve, 6) every 3 weeks plus concurrent cetuximab (400 mg/m(2) loading dose followed by 250 mg/m(2) weekly) for four cycles followed by maintenance cetuximab or sequential paclitaxel-carboplatin for four cycles followed by cetuximab.
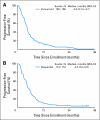
Results: Of 242 patients enrolled, 224 were eligible and assessable for response (106 and 118 patients in the concurrent and sequential arms, respectively). With a median follow-up time of 32 months, the median overall survival was 10.9 months (95% CI, 9.2 to 13.0 months) for patients receiving concurrent therapy and 10.7 months (95% CI, 8.5 to 12.8 months) for patients receiving sequential therapy (P = .57); 1-year survival rates were 45% (95% CI, 36% to 54%) and 44% (95% CI, 35% to 53%), respectively. Response rates and progression-free survival times were similar in both arms, as was grade 3 rash, whereas sensory neuropathy was higher in the concurrent arm (15% v 5% in the sequential arm; P = .036).
Conclusion: Although both regimens met the efficacy criterion for continued evaluation, the concurrent regimen of paclitaxel/carboplatin plus cetuximab was chosen.
Figures



References
-
- Kelly K, Crowley J, Bunn PA, Jr, et al. Randomized phase III trial of paclitaxel plus carboplatin versus vinorelbine plus cisplatin in the treatment of patients with advanced non-small-cell lung cancer: A Southwest Oncology Group trial. J Clin Oncol. 2001;19:3210–3218. - PubMed
-
- Schiller JH, Harrington D, Belani CP, et al. Comparison of four chemotherapy regimens for advanced non-small-cell lung cancer. N Engl J Med. 2002;346:92–98. - PubMed
-
- Sandler A, Gray R, Perry MC, et al. Paclitaxel-carboplatin alone or with bevacizumab for non-small-cell lung cancer. N Engl J Med. 2006;355:2542–2550. - PubMed
-
- Giaccone G, Herbst RS, Manegold C, et al. Gefitinib in combination with gemcitabine and cisplatin in advanced non-small-cell lung cancer: A phase III trial—INTACT 1. J Clin Oncol. 2004;22:777–784. - PubMed
-
- Herbst RS, Giaccone G, Schiller JH, et al. Gefitinib in combination with paclitaxel and carboplatin in advanced non-small-cell lung cancer: A phase III trial—INTACT 2. J Clin Oncol. 2004;22:785–794. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- N01 CA004919/CA/NCI NIH HHS/United States
- U10 CA012644/CA/NCI NIH HHS/United States
- CA22433/CA/NCI NIH HHS/United States
- U10 CA045808/CA/NCI NIH HHS/United States
- CA68183/CA/NCI NIH HHS/United States
- CA63850/CA/NCI NIH HHS/United States
- U10 CA035178/CA/NCI NIH HHS/United States
- CA76447/CA/NCI NIH HHS/United States
- U10 CA105409/CA/NCI NIH HHS/United States
- CA76448/CA/NCI NIH HHS/United States
- U10 CA046368/CA/NCI NIH HHS/United States
- U10 CA035090/CA/NCI NIH HHS/United States
- U10 CA063844/CA/NCI NIH HHS/United States
- CA63848/CA/NCI NIH HHS/United States
- CA37981/CA/NCI NIH HHS/United States
- P30 CA093373/CA/NCI NIH HHS/United States
- U10 CA022433/CA/NCI NIH HHS/United States
- CA58416/CA/NCI NIH HHS/United States
- U10 CA027057/CA/NCI NIH HHS/United States
- CA35128/CA/NCI NIH HHS/United States
- U10 CA037981/CA/NCI NIH HHS/United States
- U10 CA004919/CA/NCI NIH HHS/United States
- N01 CA035431/CA/NCI NIH HHS/United States
- U10 CA046113/CA/NCI NIH HHS/United States
- U10 CA045560/CA/NCI NIH HHS/United States
- CA12644/CA/NCI NIH HHS/United States
- U10 CA035128/CA/NCI NIH HHS/United States
- CA20319/CA/NCI NIH HHS/United States
- U10 CA063850/CA/NCI NIH HHS/United States
- U10 CA076447/CA/NCI NIH HHS/United States
- N01 CA032102/CA/NCI NIH HHS/United States
- U10 CA014028/CA/NCI NIH HHS/United States
- N01 CA035119/CA/NCI NIH HHS/United States
- CA45808/CA/NCI NIH HHS/United States
- U10 CA180846/CA/NCI NIH HHS/United States
- N01 CA046441/CA/NCI NIH HHS/United States
- U10 CA063848/CA/NCI NIH HHS/United States
- CA14028/CA/NCI NIH HHS/United States
- CA58882/CA/NCI NIH HHS/United States
- CA11083/CA/NCI NIH HHS/United States
- CA58861/CA/NCI NIH HHS/United States
- CA35090/CA/NCI NIH HHS/United States
- CA46282/CA/NCI NIH HHS/United States
- CA76132/CA/NCI NIH HHS/United States
- N01 CA063844/CA/NCI NIH HHS/United States
- U10 CA045450/CA/NCI NIH HHS/United States
- U10 CA032102/CA/NCI NIH HHS/United States
- U10 CA046282/CA/NCI NIH HHS/United States
- CA45450/CA/NCI NIH HHS/United States
- CA105409/CA/NCI NIH HHS/United States
- N01 CA035178/CA/NCI NIH HHS/United States
- CA46368/CA/NCI NIH HHS/United States
- CA67663/CA/NCI NIH HHS/United States
- N01 CA038926/CA/NCI NIH HHS/United States
- U10 CA067575/CA/NCI NIH HHS/United States
- N01 CA027057/CA/NCI NIH HHS/United States
- U10 CA046441/CA/NCI NIH HHS/United States
- UG1 CA233340/CA/NCI NIH HHS/United States
- U10 CA058882/CA/NCI NIH HHS/United States
- U10 CA020319/CA/NCI NIH HHS/United States
- CA46113/CA/NCI NIH HHS/United States
- U10 CA038926/CA/NCI NIH HHS/United States
- U10 CA042777/CA/NCI NIH HHS/United States
- U10 CA035431/CA/NCI NIH HHS/United States
- U10 CA035119/CA/NCI NIH HHS/United States
- CA42777/CA/NCI NIH HHS/United States
- U10 CA011083/CA/NCI NIH HHS/United States
- N01 CA067575/CA/NCI NIH HHS/United States
- U10 CA067663/CA/NCI NIH HHS/United States
- U10 CA058861/CA/NCI NIH HHS/United States
- U10 CA068183/CA/NCI NIH HHS/United States
- N01 CA045560/CA/NCI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

