The cargo-selective retromer complex is a recruiting hub for protein complexes that regulate endosomal tubule dynamics
- PMID: 20923837
- PMCID: PMC2964111
- DOI: 10.1242/jcs.071472
The cargo-selective retromer complex is a recruiting hub for protein complexes that regulate endosomal tubule dynamics
Abstract
The retromer complex is required for the efficient endosome-to-Golgi retrieval of the CIMPR, sortilin, SORL1, wntless and other physiologically important membrane proteins. Retromer comprises two protein complexes that act together in endosome-to-Golgi retrieval; the cargo-selective complex is a trimer of VPS35, VPS29 and VPS26 that sorts cargo into tubules for retrieval to the Golgi. Tubules are produced by the oligomerization of sorting nexin dimers. Here, we report the identification of five endosomally-localised proteins that modulate tubule formation and are recruited to the membrane via interactions with the cargo-selective retromer complex. One of the retromer-interacting proteins, strumpellin, is mutated in hereditary spastic paraplegia, a progressive length-dependent axonopathy. Here, we show that strumpellin regulates endosomal tubules as part of a protein complex with three other proteins that include WASH1, an actin-nucleating promoting factor. Therefore, in addition to a direct role in endosome-to-Golgi retrieval, the cargo-selective retromer complex also acts as a platform for recruiting physiologically important proteins to endosomal membranes that regulate membrane tubule dynamics.
Figures



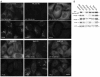






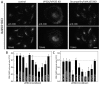
References
-
- Attar N., Cullen P. J. (2009). The retromer complex. Adv Enzyme Regul. 50, 216-236 - PubMed
-
- Bache K. G., Raiborg C., Mehlum A., Stenmark H. (2003). STAM and Hrs are subunits of a multivalent ubiquitin-binding complex on early endosomes. J. Biol. Chem. 278, 12513-12521 - PubMed
-
- Carlton J., Bujny M., Peter B. J., Oorschot V. M., Rutherford A., Mellor H., Klumperman J., McMahon H. T., Cullen P. J. (2004). Sorting nexin-1 mediates tubular endosome-to-TGN transport through coincidence sensing of high-curvature membranes and 3-phosphoinositides. Curr. Biol. 14, 1791-1800 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

