Ablation of skeletal muscle triadin impairs FKBP12/RyR1 channel interactions essential for maintaining resting cytoplasmic Ca2+
- PMID: 20926377
- PMCID: PMC2992278
- DOI: 10.1074/jbc.M110.164525
Ablation of skeletal muscle triadin impairs FKBP12/RyR1 channel interactions essential for maintaining resting cytoplasmic Ca2+
Abstract
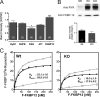
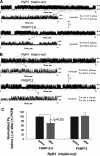
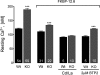
Previously, we have shown that lack of expression of triadins in skeletal muscle cells results in significant increase of myoplasmic resting free Ca(2+) ([Ca(2+)](rest)), suggesting a role for triadins in modulating global intracellular Ca(2+) homeostasis. To understand this mechanism, we study here how triadin alters [Ca(2+)](rest), Ca(2+) release, and Ca(2+) entry pathways using a combination of Ca(2+) microelectrodes, channels reconstituted in bilayer lipid membranes (BLM), Ca(2+), and Mn(2+) imaging analyses of myotubes and RyR1 channels obtained from triadin-null mice. Unlike WT cells, triadin-null myotubes had chronically elevated [Ca(2+)](rest) that was sensitive to inhibition with ryanodine, suggesting that triadin-null cells have increased basal RyR1 activity. Consistently, BLM studies indicate that, unlike WT-RyR1, triadin-null channels more frequently display atypical gating behavior with multiple and stable subconductance states. Accordingly, pulldown analysis and fluorescent FKBP12 binding studies in triadin-null muscles revealed a significant impairment of the FKBP12/RyR1 interaction. Mn(2+) quench rates under resting conditions indicate that triadin-null cells also have higher Ca(2+) entry rates and lower sarcoplasmic reticulum Ca(2+) load than WT cells. Overexpression of FKBP12.6 reverted the null phenotype, reducing resting Ca(2+) entry, recovering sarcoplasmic reticulum Ca(2+) content levels, and restoring near normal [Ca(2+)](rest). Exogenous FKBP12.6 also reduced the RyR1 channel P(o) but did not rescue subconductance behavior. In contrast, FKBP12 neither reduced P(o) nor recovered multiple subconductance gating. These data suggest that elevated [Ca(2+)](rest) in triadin-null myotubes is primarily driven by dysregulated RyR1 channel activity that results in part from impaired FKBP12/RyR1 functional interactions and a secondary increased Ca(2+) entry at rest.
Figures



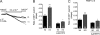
Similar articles
-
Reduced gain of excitation-contraction coupling in triadin-null myotubes is mediated by the disruption of FKBP12/RyR1 interaction.Cell Calcium. 2011 Feb;49(2):128-35. doi: 10.1016/j.ceca.2011.01.005. Cell Calcium. 2011. PMID: 21310482 Free PMC article.
-
Triadin binding to the C-terminal luminal loop of the ryanodine receptor is important for skeletal muscle excitation contraction coupling.J Gen Physiol. 2007 Oct;130(4):365-78. doi: 10.1085/jgp.200709790. Epub 2007 Sep 10. J Gen Physiol. 2007. PMID: 17846166 Free PMC article.
-
Occurrence of atypical Ca2+ transients in triadin-binding deficient-RYR1 mutants.Biochem Biophys Res Commun. 2006 Dec 29;351(4):909-14. doi: 10.1016/j.bbrc.2006.10.115. Epub 2006 Nov 7. Biochem Biophys Res Commun. 2006. PMID: 17092484
-
Control of muscle ryanodine receptor calcium release channels by proteins in the sarcoplasmic reticulum lumen.Clin Exp Pharmacol Physiol. 2009 Mar;36(3):340-5. doi: 10.1111/j.1440-1681.2008.05094.x. Clin Exp Pharmacol Physiol. 2009. PMID: 19278523 Review.
-
Core skeletal muscle ryanodine receptor calcium release complex.Clin Exp Pharmacol Physiol. 2017 Jan;44(1):3-12. doi: 10.1111/1440-1681.12676. Clin Exp Pharmacol Physiol. 2017. PMID: 27696487 Review.
Cited by
-
On the footsteps of Triadin and its role in skeletal muscle.World J Biol Chem. 2011 Aug 26;2(8):177-83. doi: 10.4331/wjbc.v2.i8.177. World J Biol Chem. 2011. PMID: 21909459 Free PMC article.
-
Myoplasmic resting Ca2+ regulation by ryanodine receptors is under the control of a novel Ca2+-binding region of the receptor.Biochem J. 2014 Jun 1;460(2):261-71. doi: 10.1042/BJ20131553. Biochem J. 2014. PMID: 24635445 Free PMC article.
-
Absence of triadin, a protein of the calcium release complex, is responsible for cardiac arrhythmia with sudden death in human.Hum Mol Genet. 2012 Jun 15;21(12):2759-67. doi: 10.1093/hmg/dds104. Epub 2012 Mar 14. Hum Mol Genet. 2012. PMID: 22422768 Free PMC article.
-
Calcium entry units (CEUs): perspectives in skeletal muscle function and disease.J Muscle Res Cell Motil. 2021 Jun;42(2):233-249. doi: 10.1007/s10974-020-09586-3. Epub 2020 Aug 18. J Muscle Res Cell Motil. 2021. PMID: 32812118 Free PMC article.
-
Triadin/Junctin double null mouse reveals a differential role for Triadin and Junctin in anchoring CASQ to the jSR and regulating Ca(2+) homeostasis.PLoS One. 2012;7(7):e39962. doi: 10.1371/journal.pone.0039962. Epub 2012 Jul 2. PLoS One. 2012. PMID: 22768324 Free PMC article.
References
-
- Buck E. D., Nguyen H. T., Pessah I. N., Allen P. D. (1997) J. Biol. Chem. 272, 7360–7367 - PubMed
-
- Lopez J. R., Alamo L. A., Jones D. E., Papp L., Allen P. D., Gergely J., Sréter F. A. (1986) Muscle Nerve 9, 85–86 - PubMed
-
- Yang T., Esteve E., Pessah I. N., Molinski T. F., Allen P. D., López J. R. (2007) Am. J. Physiol. Cell. Physiol. 292, C1591–1598 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

