Long-term MR cell tracking of neural stem cells grafted in immunocompetent versus immunodeficient mice reveals distinct differences in contrast between live and dead cells
- PMID: 20928883
- PMCID: PMC3031985
- DOI: 10.1002/mrm.22613
Long-term MR cell tracking of neural stem cells grafted in immunocompetent versus immunodeficient mice reveals distinct differences in contrast between live and dead cells
Abstract
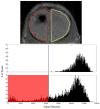
Neural stem cell (NSC)-based therapy is actively being pursued in preclinical and clinical disease models. Magnetic resonance imaging (MRI) cell tracking promises to optimize current cell transplantation paradigms, however, it is limited by dilution of contrast agent during cellular proliferation, transfer of label from dying cells to surrounding endogenous host cells, and/or biodegradation of the label. Here, we evaluated the applicability of magnetic resonance imaging for long-term tracking of transplanted neural stem cells labeled with superparamagnetic iron oxide and transfected with the bioluminescence reporter gene luciferase. Mouse neural stem cells were transplanted into immunodeficient, graft-accepting Rag2 mice or immunocompetent, graft-rejecting Balb/c mice. Hypointense voxel signals and bioluminescence were monitored over a period of 93 days. Unexpectedly, in mice that rejected the cells, the hypointense MR signal persisted throughout the entire time-course, whereas in the nonrejecting mice, the contrast cleared at a faster rate. In immunocompetent, graft-rejecting Balb/c mice, infiltrating leukocytes, and microglia were found surrounding dead cells and internalizing superparamagnetic iron oxide clusters. The present results indicate that live cell proliferation and associated label dilution may dominate contrast clearance as compared with cell death and subsequent transfer and retention of superparamagnetic iron oxide within phagocytes and brain interstitium. Thus, interpretation of signal changes during long-term MR cell tracking is complex and requires caution.
Copyright © 2010 Wiley-Liss, Inc.
Figures






References
-
- Komitova M, Mattsson B, Johansson BB, Eriksson PS. Enriched environment increases neural stem/progenitor cell proliferation and neurogenesis in the subventricular zone of stroke-lesioned adult rats. Stroke. 2005;36:1278–1282. - PubMed
-
- Obermair FJ, Schroter A, Thallmair M. Endogenous neural progenitor cells as therapeutic target after spinal cord injury. Physiology (Bethesda) 2008;23:296–304. - PubMed
-
- Ben-Hur T, van Heeswijk RB, Einstein O, Aharonowiz M, Xue R, Frost EE, Mori S, Reubinoff BE, Bulte JW. Serial in vivo MR tracking of magnetically labeled neural spheres transplanted in chronic EAE mice. Magn Reson Med. 2007;57:164–171. - PubMed
-
- Walczak P, Bulte JW. The role of noninvasive cellular imaging in developing cell-based therapies for neurodegenerative disorders. Neurodegener Dis. 2007;4:306–313. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

