Probing the spontaneous membrane insertion of a tail-anchored membrane protein by sum frequency generation spectroscopy
- PMID: 20932011
- PMCID: PMC2965819
- DOI: 10.1021/ja106508f
Probing the spontaneous membrane insertion of a tail-anchored membrane protein by sum frequency generation spectroscopy
Abstract
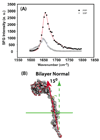
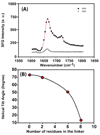
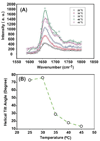
In addition to providing a semipermeable barrier that protects a cell from harmful stimuli, lipid membranes occupy a central role in hosting a variety of biological processes, including cellular communications and membrane protein functions. Most importantly, protein-membrane interactions are implicated in a variety of diseases and therefore many analytical techniques were developed to study the basis of these interactions and their influence on the molecular architecture of the cell membrane. In this study, sum frequency generation (SFG) vibrational spectroscopy is used to investigate the spontaneous membrane insertion process of cytochrome b(5) and its mutants. Experimental results show a significant difference in the membrane insertion and orientation properties of these proteins, which can be correlated with their functional differences. In particular, our results correlate the nonfunctional property of a mutant cytochrome b(5) with its inability to insert into the lipid bilayer. The approach reported in this study could be used as a potential rapid screening tool in measuring the topology of membrane proteins as well as interactions of biomolecules with lipid bilayers in situ.
Figures
Similar articles
-
Effects of membrane mimetics on cytochrome P450-cytochrome b5 interactions characterized by NMR spectroscopy.J Biol Chem. 2015 May 15;290(20):12705-18. doi: 10.1074/jbc.M114.597096. Epub 2015 Mar 20. J Biol Chem. 2015. PMID: 25795780 Free PMC article.
-
Transmembrane Interactions of Full-length Mammalian Bitopic Cytochrome-P450-Cytochrome-b5 Complex in Lipid Bilayers Revealed by Sensitivity-Enhanced Dynamic Nuclear Polarization Solid-state NMR Spectroscopy.Sci Rep. 2017 Jun 23;7(1):4116. doi: 10.1038/s41598-017-04219-1. Sci Rep. 2017. PMID: 28646173 Free PMC article.
-
The carboxyl terminus of the membrane-binding domain of cytochrome b5 spans the bilayer of the endoplasmic reticulum.J Biol Chem. 1995 Feb 17;270(7):3414-22. doi: 10.1074/jbc.270.7.3414. J Biol Chem. 1995. PMID: 7852428
-
In situ molecular level studies on membrane related peptides and proteins in real time using sum frequency generation vibrational spectroscopy.J Struct Biol. 2009 Oct;168(1):61-77. doi: 10.1016/j.jsb.2009.03.006. Epub 2009 Mar 21. J Struct Biol. 2009. PMID: 19306928 Free PMC article. Review.
-
Tethered bilayer lipid membranes (tBLMs): interest and applications for biological membrane investigations.Biochimie. 2014 Dec;107 Pt A:135-42. doi: 10.1016/j.biochi.2014.06.021. Epub 2014 Jul 4. Biochimie. 2014. PMID: 24998327 Review.
Cited by
-
Physiologically-relevant modes of membrane interactions by the human antimicrobial peptide, LL-37, revealed by SFG experiments.Sci Rep. 2013;3:1854. doi: 10.1038/srep01854. Sci Rep. 2013. PMID: 23676762 Free PMC article.
-
Evidence of a molecular boundary lubricant at snakeskin surfaces.J R Soc Interface. 2015 Dec 6;12(113):20150817. doi: 10.1098/rsif.2015.0817. J R Soc Interface. 2015. PMID: 26655468 Free PMC article.
-
Single Lipid Bilayers Constructed on Polymer Cushion Studied by Sum Frequency Generation Vibrational Spectroscopy.J Phys Chem C Nanomater Interfaces. 2011 Apr 21;115(15):7613-7620. doi: 10.1021/jp200546h. J Phys Chem C Nanomater Interfaces. 2011. PMID: 21712964 Free PMC article.
-
Membrane orientation of MSI-78 measured by sum frequency generation vibrational spectroscopy.Langmuir. 2011 Jun 21;27(12):7760-7. doi: 10.1021/la201048t. Epub 2011 May 19. Langmuir. 2011. PMID: 21595453 Free PMC article.
-
Dependence of Alamethicin Membrane Orientation on the Solution Concentration.J Phys Chem C Nanomater Interfaces. 2013 Feb 21;117(7):3358-3365. doi: 10.1021/jp3099522. Epub 2013 Jan 24. J Phys Chem C Nanomater Interfaces. 2013. PMID: 23565299 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

