PLCγ-activated signalling is essential for TrkB mediated sensory neuron structural plasticity
- PMID: 20932311
- PMCID: PMC2964534
- DOI: 10.1186/1471-213X-10-103
PLCγ-activated signalling is essential for TrkB mediated sensory neuron structural plasticity
Abstract
Background: The vestibular system provides the primary input of our sense of balance and spatial orientation. Dysfunction of the vestibular system can severely affect a person's quality of life. Therefore, understanding the molecular basis of vestibular neuron survival, maintenance, and innervation of the target sensory epithelia is fundamental.
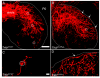
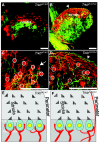
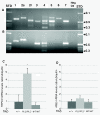
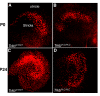
Results: Here we report that a point mutation at the phospholipase Cγ (PLCγ) docking site in the mouse neurotrophin tyrosine kinase receptor TrkB (Ntrk2) specifically impairs fiber guidance inside the vestibular sensory epithelia, but has limited effects on the survival of vestibular sensory neurons and growth of afferent processes toward the sensory epithelia. We also show that expression of the TRPC3 cation calcium channel, whose activity is known to be required for nerve-growth cone guidance induced by brain-derived neurotrophic factor (BDNF), is altered in these animals. In addition, we find that absence of the PLCγ mediated TrkB signalling interferes with the transformation of bouton type afferent terminals of vestibular dendrites into calyces (the largest synaptic contact of dendrites known in the mammalian nervous system) on type I vestibular hair cells; the latter are normally distributed in these mutants as revealed by an unaltered expression pattern of the potassium channel KCNQ4 in these cells.
Conclusions: These results demonstrate a crucial involvement of the TrkB/PLCγ-mediated intracellular signalling in structural aspects of sensory neuron plasticity.
Figures

Similar articles
-
Developmental expression of Kcnq4 in vestibular neurons and neurosensory epithelia.Brain Res. 2007 Mar 30;1139:117-25. doi: 10.1016/j.brainres.2006.12.087. Epub 2007 Jan 8. Brain Res. 2007. PMID: 17292869 Free PMC article.
-
BDNF gene replacement reveals multiple mechanisms for establishing neurotrophin specificity during sensory nervous system development.Development. 2003 Apr;130(8):1479-91. doi: 10.1242/dev.00378. Development. 2003. PMID: 12620975
-
In vivo and in vitro localization of brain-derived neurotrophic factor, fibroblast growth factor-2 and their receptors in the bullfrog vestibular end organs.Brain Res Mol Brain Res. 2002 Jun 15;102(1-2):83-99. doi: 10.1016/s0169-328x(02)00202-4. Brain Res Mol Brain Res. 2002. PMID: 12191497
-
BDNF-induced local protein synthesis and synaptic plasticity.Neuropharmacology. 2014 Jan;76 Pt C:639-56. doi: 10.1016/j.neuropharm.2013.04.005. Epub 2013 Apr 16. Neuropharmacology. 2014. PMID: 23602987 Review.
-
Neurotrophins in the ear: their roles in sensory neuron survival and fiber guidance.Prog Brain Res. 2004;146:265-78. doi: 10.1016/S0079-6123(03)46017-2. Prog Brain Res. 2004. PMID: 14699969 Review.
Cited by
-
The role of rab proteins in neuronal cells and in the trafficking of neurotrophin receptors.Membranes (Basel). 2014 Oct 6;4(4):642-77. doi: 10.3390/membranes4040642. Membranes (Basel). 2014. PMID: 25295627 Free PMC article. Review.
-
The molecular basis of making spiral ganglion neurons and connecting them to hair cells of the organ of Corti.Hear Res. 2011 Aug;278(1-2):21-33. doi: 10.1016/j.heares.2011.03.002. Epub 2011 Mar 21. Hear Res. 2011. PMID: 21414397 Free PMC article. Review.
-
Activation of Three Major Signaling Pathways After Endurance Training and Spinal Cord Injury.Mol Neurobiol. 2022 Feb;59(2):950-967. doi: 10.1007/s12035-021-02628-y. Epub 2021 Nov 22. Mol Neurobiol. 2022. PMID: 34811634 Free PMC article.
-
Inner ear hair cells deteriorate in mice engineered to have no or diminished innervation.Front Aging Neurosci. 2015 Mar 18;7:33. doi: 10.3389/fnagi.2015.00033. eCollection 2015. Front Aging Neurosci. 2015. PMID: 25852547 Free PMC article.
-
Long-term Administration of Salicylate-induced Changes in BDNF Expression and CREB Phosphorylation in the Auditory Cortex of Rats.Otol Neurotol. 2018 Mar;39(3):e173-e180. doi: 10.1097/MAO.0000000000001717. Otol Neurotol. 2018. PMID: 29342042 Free PMC article.
References
-
- Lysakowski AL, Goldberg JM. In: The Anatomy and Physiology of the Central and Peripheral Vestibular System. Highstein S, Popper AN, Fay RR, editor. New York: Springer; 2003. Morphophysiology of the vestibular periphery; pp. 57–152.
-
- Spoendlin H. In: In Physiology of the ear. Jahn A, Santos-Sachi J, editor. New York: Raven Press; 1988. Neural anatomy of the inner ear; pp. 201–219.
-
- Fritzsch B, Tessarollo L, Coppola V, Reichardt LF. Neurotrophins in the ear: their roles in sensory neuron survival and fiber guidance. Prog Brain Res. 2004;146:265–278. full_text. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

