CXCL10 promotes liver fibrosis by prevention of NK cell mediated hepatic stellate cell inactivation
- PMID: 20932719
- PMCID: PMC3855675
- DOI: 10.1016/j.jaut.2010.09.003
CXCL10 promotes liver fibrosis by prevention of NK cell mediated hepatic stellate cell inactivation
Abstract
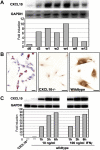
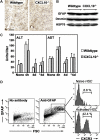
Chemokines, such as CXCL10, promote hepatic inflammation in chronic or acute liver injury through recruitment of leukocytes to the liver parenchyma. The CXCL10 receptor CXCR3, which is expressed on a subset of leukocytes, plays an important part in Th1-dependent inflammatory responses. Here, we investigated the role of CXCL10 in chemically induced liver fibrosis. We used carbon tetrachloride (CCl(4)) to trigger chronic liver damage in wildtype C57BL/6 and CXCL10-deficient mice. Fibrosis severity was assessed by Sirius Red staining and intrahepatic leukocyte subsets were investigated by immunohistochemistry. We have further analyzed hepatic stellate cell (HSC) distribution and activation and investigated the effect of CXCL10 on HSC motility and proliferation. In order to demonstrate a possible therapeutic intervention strategy, we have examined the anti-fibrotic potential of a neutralizing anti-CXCL10 antibody. Upon CCl(4) administration, CXCL10-deficient mice showed massively reduced liver fibrosis, when compared to wildtype mice. CXCL10-deficient mice had less B- and T lymphocyte and dendritic cell infiltrations within the liver and the number and activity of HSCs was reduced. In contrast, natural killer (NK) cells were more abundant in CXCL10-deficient mice and granzyme B expression was increased in areas with high numbers of NK cells. Further detailed analysis revealed that HSCs express CXCR3, respond to CXCL10 and secrete CXCL10 when stimulated with IFNγ. Blockade of CXCL10 with a neutralizing antibody exhibited a significant anti-fibrotic effect. Our data suggest that CXCL10 is a pro-fibrotic factor, which participates in a crosstalk between hepatocytes, HSCs and immune cells. NK cells seem to play an important role in controlling HSC activity and fibrosis. CXCL10 blockade may constitute a possible therapeutic intervention for hepatic fibrosis.
Copyright © 2010 Elsevier Ltd. All rights reserved.
Figures





Similar articles
-
The CXCR3(+)CD56Bright phenotype characterizes a distinct NK cell subset with anti-fibrotic potential that shows dys-regulated activity in hepatitis C.PLoS One. 2012;7(7):e38846. doi: 10.1371/journal.pone.0038846. Epub 2012 Jul 5. PLoS One. 2012. PMID: 22792160 Free PMC article.
-
Antifibrotic effects of CXCL9 and its receptor CXCR3 in livers of mice and humans.Gastroenterology. 2009 Jul;137(1):309-19, 319.e1-3. doi: 10.1053/j.gastro.2009.03.053. Epub 2009 Apr 1. Gastroenterology. 2009. PMID: 19344719 Free PMC article.
-
Interleukin-27 and IFNγ regulate the expression of CXCL9, CXCL10, and CXCL11 in hepatitis.J Mol Med (Berl). 2015 Dec;93(12):1355-67. doi: 10.1007/s00109-015-1319-6. Epub 2015 Jul 23. J Mol Med (Berl). 2015. PMID: 26199110
-
Cell death and fibrogenesis.Semin Liver Dis. 2010 Aug;30(3):226-31. doi: 10.1055/s-0030-1255352. Epub 2010 Jul 21. Semin Liver Dis. 2010. PMID: 20665375 Free PMC article. Review.
-
Crosstalk between hepatic stellate cells and surrounding cells in hepatic fibrosis.Int Immunopharmacol. 2021 Oct;99:108051. doi: 10.1016/j.intimp.2021.108051. Epub 2021 Aug 18. Int Immunopharmacol. 2021. PMID: 34426110 Review.
Cited by
-
MyD88 in hepatic stellate cells enhances liver fibrosis via promoting macrophage M1 polarization.Cell Death Dis. 2022 Apr 28;13(4):411. doi: 10.1038/s41419-022-04802-z. Cell Death Dis. 2022. PMID: 35484116 Free PMC article.
-
Dynamic YAP expression in the non-parenchymal liver cell compartment controls heterologous cell communication.Cell Mol Life Sci. 2024 Mar 4;81(1):115. doi: 10.1007/s00018-024-05126-1. Cell Mol Life Sci. 2024. PMID: 38436764 Free PMC article.
-
Characterisation of LPS+ bacterial extracellular vesicles along the gut-hepatic portal vein-liver axis.J Extracell Vesicles. 2024 Jul;13(7):e12474. doi: 10.1002/jev2.12474. J Extracell Vesicles. 2024. PMID: 39001704 Free PMC article.
-
Exploration of immune infiltration and feature genes in viral hepatitis-associated liver fibrosis using transcriptome data.Ann Transl Med. 2022 Oct;10(19):1051. doi: 10.21037/atm-22-2205. Ann Transl Med. 2022. PMID: 36330414 Free PMC article.
-
Primary biliary cirrhosis: From bench to bedside.World J Gastrointest Pharmacol Ther. 2015 Aug 6;6(3):32-58. doi: 10.4292/wjgpt.v6.i3.32. World J Gastrointest Pharmacol Ther. 2015. PMID: 26261733 Free PMC article. Review.
References
-
- Friedman SL, Rockey DC, Bissell DM. Hepatic fibrosis 2006: report of the Third AASLD Single Topic Conference. Hepatology. 2007;45:242–9. - PubMed
-
- Cheung O, Sanyal AJ. Recent advances in nonalcoholic fatty liver disease. Curr Opin Gastroenterol. 2009;25:230–7. - PubMed
-
- Czaja AJ. Autoimmune liver disease. Curr Opin Gastroenterol. 2009;25:215–22. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

