Escherichia coli binary culture engineered for direct fermentation of hemicellulose to a biofuel
- PMID: 20935118
- PMCID: PMC3008245
- DOI: 10.1128/AEM.00908-10
Escherichia coli binary culture engineered for direct fermentation of hemicellulose to a biofuel
Abstract
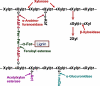
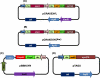
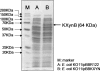
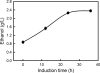
Metabolic engineering has created several Escherichia coli biocatalysts for production of biofuels and other useful molecules. However, the inability of these biocatalysts to directly use polymeric substrates necessitates costly pretreatment and enzymatic hydrolysis prior to fermentation. Consolidated bioprocessing has the potential to simplify the process by combining enzyme production, hydrolysis, and fermentation into a single step but requires a fermenting organism to multitask by producing both necessary enzymes and target molecules. We demonstrate here a binary strategy for consolidated bioprocessing of xylan, a complex substrate requiring six hemicellulases for complete hydrolysis. An integrated modular approach was used to design the two strains to function cooperatively in the process of transforming xylan into ethanol. The first strain was engineered to coexpress two hemicellulases. Recombinant enzymes were secreted to the growth medium by a method of lpp deletion with over 90% efficiency. Secreted enzymes hydrolyzed xylan into xylooligosaccharides, which were taken in by the second strain, designed to use the xylooligosaccharides for ethanol production. Cocultivation of the two strains converted xylan hemicellulose to ethanol with a yield about 55% of the theoretical value. Inclusion of other three hemicellulases improved the ethanol yield to 70%. Analysis of the culture broth showed that xylooligosaccharides with four or more xylose units were not utilized, suggesting that improving the use of higher xyloogligomers should be the focus in future efforts. This is the first demonstration of an engineered binary culture for consolidated bioprocessing of xylan. The modular design should allow the strategy to be adopted for a broad range of biofuel and biorefinery products.
Figures


Similar articles
-
Engineering Escherichia coli for succinate production from hemicellulose via consolidated bioprocessing.Microb Cell Fact. 2012 Mar 29;11:37. doi: 10.1186/1475-2859-11-37. Microb Cell Fact. 2012. PMID: 22455836 Free PMC article.
-
Conversion of xylan to ethanol by ethanologenic strains of Escherichia coli and Klebsiella oxytoca.Appl Environ Microbiol. 1992 Apr;58(4):1128-33. doi: 10.1128/aem.58.4.1128-1133.1992. Appl Environ Microbiol. 1992. PMID: 1599236 Free PMC article.
-
Direct conversion of xylan to ethanol by recombinant Saccharomyces cerevisiae strains displaying an engineered minihemicellulosome.Appl Environ Microbiol. 2012 Jun;78(11):3837-45. doi: 10.1128/AEM.07679-11. Epub 2012 Mar 23. Appl Environ Microbiol. 2012. PMID: 22447594 Free PMC article.
-
Polysaccharide hydrolysis with engineered Escherichia coli for the production of biocommodities.J Ind Microbiol Biotechnol. 2013 May;40(5):401-10. doi: 10.1007/s10295-013-1245-y. Epub 2013 Mar 12. J Ind Microbiol Biotechnol. 2013. PMID: 23478881 Review.
-
Smart fermentation engineering for butanol production: designed biomass and consolidated bioprocessing systems.Appl Microbiol Biotechnol. 2019 Dec;103(23-24):9359-9371. doi: 10.1007/s00253-019-10198-2. Epub 2019 Nov 13. Appl Microbiol Biotechnol. 2019. PMID: 31720773 Review.
Cited by
-
Understanding the Mechanisms of Positive Microbial Interactions That Benefit Lactic Acid Bacteria Co-cultures.Front Microbiol. 2020 Sep 4;11:2088. doi: 10.3389/fmicb.2020.02088. eCollection 2020. Front Microbiol. 2020. PMID: 33013761 Free PMC article. Review.
-
Downregulation of GAUT12 in Populus deltoides by RNA silencing results in reduced recalcitrance, increased growth and reduced xylan and pectin in a woody biofuel feedstock.Biotechnol Biofuels. 2015 Mar 12;8:41. doi: 10.1186/s13068-015-0218-y. eCollection 2015. Biotechnol Biofuels. 2015. PMID: 25802552 Free PMC article.
-
Biorefining of protein waste for production of sustainable fuels and chemicals.Biotechnol Biofuels. 2018 Sep 20;11:256. doi: 10.1186/s13068-018-1234-5. eCollection 2018. Biotechnol Biofuels. 2018. PMID: 30250508 Free PMC article. Review.
-
Spontaneously established syntrophic yeast communities improve bioproduction.Nat Chem Biol. 2023 Aug;19(8):951-961. doi: 10.1038/s41589-023-01341-2. Epub 2023 May 29. Nat Chem Biol. 2023. PMID: 37248413 Free PMC article.
-
Recent advances in microbial co-culture for production of value-added compounds.3 Biotech. 2022 May;12(5):115. doi: 10.1007/s13205-022-03177-4. Epub 2022 Apr 19. 3 Biotech. 2022. PMID: 35547018 Free PMC article. Review.
References
-
- Carvalheiro, F., L. C. Duarte, and F. M. Girio. 2008. Hemicellulose biorefineries: a review on biomass pretreatments. J. Sci. Ind. Res. 67:849-864.
-
- Chaplin, M. F. 1986. Monosaccharides, p. 1-36. In M. F. Chaplin and J. F. Kennedy (ed.), Carbohydrate analysis: a practical approach. IRL Press Oxford, Oxford, United Kingdom.
-
- D'Urso, A., R. Bubbico, M. Scarsella, and M. Bravi. 2008. Fungal biomass-based production of lipids for biodiesel synthesis. Chem. Eng. Trans. 14:251-258.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

