PA-824 exhibits time-dependent activity in a murine model of tuberculosis
- PMID: 20937781
- PMCID: PMC3019674
- DOI: 10.1128/AAC.00849-10
PA-824 exhibits time-dependent activity in a murine model of tuberculosis
Abstract
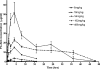
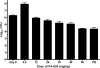
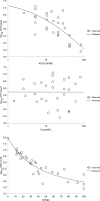
PA-824 is one of two nitroimidazoles in phase II clinical trials to treat tuberculosis. In mice, it has dose-dependent early bactericidal and sterilizing activity. In humans with tuberculosis, PA-824 demonstrated early bactericidal activity (EBA) at doses ranging from 200 to 1,200 mg per day, but no dose-response effect was observed. To better understand the relationship between drug exposure and effect, we performed a dose fractionation study in mice. Dose-ranging pharmacokinetic data were used to simulate drug exposure profiles. Beginning 2 weeks after aerosol infection with Mycobacterium tuberculosis, total PA-824 doses from 144 to 4,608 mg/kg were administered as 3, 4, 8, 12, 24, or 48 divided doses over 24 days. Lung CFU counts after treatment were strongly correlated with the free drug T(>MIC) (R(2) = 0.87) and correlated with the free drug AUC/MIC (R(2) = 0.60), but not with the free drug C(max)/MIC (R(2) = 0.17), where T(>MIC) is the cumulative percentage of the dosing interval that the drug concentration exceeds the MIC under steady-state pharmacokinetic conditions and AUC is the area under the concentration-time curve. When the data set was limited to regimens with dosing intervals of ≤72 h, both the T(>MIC) and the AUC/MIC values fit the data well. Free drug T(>MIC) of 22, 48, and 77% were associated with bacteriostasis, a 1-log kill, and a 1.59-log kill (or 80% of the maximum observed effect), respectively. Human pharmacodynamic simulations based on phase I data predict 200 mg/day produces free drug T(>MIC) values near the target for maximal observed bactericidal effect. The results support the recently demonstrated an EBA of 200 mg/day and the lack of a dose-response between 200 and 1,200 mg/day. T(>MIC), in conjunction with AUC/MIC, is the parameter on which dose optimization of PA-824 should be based.
Figures
References
-
- Balganesh, T. S., P. M. Alzari, and S. T. Cole. 2008. Rising standards for tuberculosis drug development. Trends Pharmacol. Sci. 29:576-581. - PubMed
-
- Craig, W. A. 1998. Pharmacokinetic/pharmacodynamic parameters: rationale for antibacterial dosing of mice and men. Clin. Infect. Dis. 26:1-10. - PubMed
-
- Craig, W. A. 2001. Does the dose matter? Clin. Infect. Dis. 33:S233-S237. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical