Induction of Toll-like receptor 3-mediated immunity during gestation inhibits cortical neurogenesis and causes behavioral disturbances
- PMID: 20941330
- PMCID: PMC2953007
- DOI: 10.1128/mBio.00176-10
Induction of Toll-like receptor 3-mediated immunity during gestation inhibits cortical neurogenesis and causes behavioral disturbances
Abstract
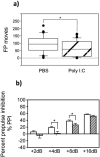
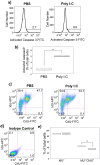
Maternal infection during pregnancy with a wide range of RNA and DNA viruses is associated with increased risk for schizophrenia and autism in their offspring. A common feature in these exposures is that virus replication induces innate immunity through interaction with Toll-like receptors (TLRs). We employed a mouse model wherein pregnant mice were exposed to polyinosinic-polycytidylic acid [poly(I ⋅ C)], a synthetic, double-stranded RNA molecular mimic of replicating virus. Poly(I ⋅ C) inhibited embryonic neuronal stem cell replication and population of the superficial layers of the neocortex by neurons. Poly(I ⋅ C) also led to impaired neonatal locomotor development and abnormal sensorimotor gating responses in adult offspring. Using Toll-like receptor 3 (TLR3)-deficient mice, we established that these effects were dependent on TLR3. Inhibition of stem cell proliferation was also abrogated by pretreatment with the nonsteroidal anti-inflammatory drug (NSAID) carprofen, a cyclooxygenase (COX) inhibitor. Our findings provide insights into mechanisms by which maternal infection can induce subtle neuropathology and behavioral dysfunction, and they may suggest strategies for reducing the risk of neuropsychiatric disorders subsequent to prenatal exposures to pathogens and other triggers of innate immunity.
Figures



Similar articles
-
Maternal host responses to poly(I:C) during pregnancy leads to both dysfunctional immune profiles and altered behaviour in the offspring.Am J Reprod Immunol. 2020 Aug;84(2):e13260. doi: 10.1111/aji.13260. Epub 2020 May 26. Am J Reprod Immunol. 2020. PMID: 32365239
-
Adult brain and behavioral pathological markers of prenatal immune challenge during early/middle and late fetal development in mice.Brain Behav Immun. 2008 May;22(4):469-86. doi: 10.1016/j.bbi.2007.09.012. Epub 2007 Nov 26. Brain Behav Immun. 2008. PMID: 18023140
-
Distinct trans-placental effects of maternal immune activation by TLR3 and TLR7 agonists: implications for schizophrenia risk.Sci Rep. 2021 Dec 13;11(1):23841. doi: 10.1038/s41598-021-03216-9. Sci Rep. 2021. PMID: 34903784 Free PMC article.
-
To poly(I:C) or not to poly(I:C): advancing preclinical schizophrenia research through the use of prenatal immune activation models.Neuropharmacology. 2012 Mar;62(3):1308-21. doi: 10.1016/j.neuropharm.2011.01.009. Epub 2011 Jan 14. Neuropharmacology. 2012. PMID: 21238465 Review.
-
The poly(I:C)-induced maternal immune activation model in preclinical neuropsychiatric drug discovery.Pharmacol Ther. 2015 May;149:213-26. doi: 10.1016/j.pharmthera.2015.01.001. Epub 2015 Jan 4. Pharmacol Ther. 2015. PMID: 25562580 Review.
Cited by
-
Role of the Toll‑like receptor 3 signaling pathway in the neuroprotective effect of sevoflurane pre‑conditioning during cardiopulmonary bypass in rats.Mol Med Rep. 2015 Dec;12(6):7859-68. doi: 10.3892/mmr.2015.4420. Epub 2015 Oct 9. Mol Med Rep. 2015. PMID: 26460219 Free PMC article.
-
Maternal Immune Activation and Schizophrenia-Evidence for an Immune Priming Disorder.Front Psychiatry. 2021 Feb 17;12:585742. doi: 10.3389/fpsyt.2021.585742. eCollection 2021. Front Psychiatry. 2021. PMID: 33679468 Free PMC article. Review.
-
RNase A Promotes Proliferation of Neuronal Progenitor Cells via an ERK-Dependent Pathway.Front Mol Neurosci. 2018 Nov 26;11:428. doi: 10.3389/fnmol.2018.00428. eCollection 2018. Front Mol Neurosci. 2018. PMID: 30534052 Free PMC article.
-
Immune-mediated animal models of Tourette syndrome.Neurosci Biobehav Rev. 2013 Jul;37(6):1120-38. doi: 10.1016/j.neubiorev.2013.01.007. Epub 2013 Jan 10. Neurosci Biobehav Rev. 2013. PMID: 23313649 Free PMC article. Review.
-
Stereotypical alterations in cortical patterning are associated with maternal illness-induced placental dysfunction.J Neurosci. 2013 Oct 23;33(43):16874-88. doi: 10.1523/JNEUROSCI.4654-12.2013. J Neurosci. 2013. PMID: 24155294 Free PMC article.
References
-
- Patterson P. H. 2007. Neuroscience. Maternal effects on schizophrenia risk. Science 318:576–577 - PubMed
-
- Schmitz C., van Kooten I. A., Hof P. R., van Engeland H., Patterson P. H., Steinbusch H. W. 2005. Autism: neuropathology, alterations of the GABAergic system, and animal models. Int. Rev. Neurobiol. 71:1–26 - PubMed
-
- Meyer U., Engler A., Weber L., Schedlowski M., Feldon J. 2008. Preliminary evidence for a modulation of fetal dopaminergic development by maternal immune activation during pregnancy. Neuroscience 154:701–709 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

