Real-time hydrogen/deuterium exchange kinetics via supercharged electrospray ionization tandem mass spectrometry
- PMID: 20942406
- PMCID: PMC3049191
- DOI: 10.1021/ac101957x
Real-time hydrogen/deuterium exchange kinetics via supercharged electrospray ionization tandem mass spectrometry
Abstract
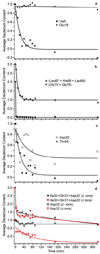
Amide hydrogen/deuterium exchange (HDX) rate constants of bovine ubiquitin in an ammonium acetate solution containing 1% of the electrospray ionization (ESI) "supercharging" reagent m-nitrobenzyl alcohol (m-NBA) were obtained using top-down, electron transfer dissociation (ETD) tandem mass spectrometry (MS). The supercharging reagent replaces the acid and temperature "quench" step in the conventional MS approach to HDX experiments by causing rapid protein denaturation to occur in the ESI droplet. The higher charge state ions that are produced with m-NBA are more unfolded, as measured by ion mobility, and result in higher fragmentation efficiency and higher sequence coverage with ETD. Single amino acid resolution was obtained for 44 of 72 exchangeable amide sites, and summed kinetic data were obtained for regions of the protein where adjacent fragment ions were not observed, resulting in an overall spatial resolution of 1.3 residues. Comparison of these results with previous values from NMR indicates that the supercharging reagent does not cause significant structural changes to the protein in the initial ESI solution and that scrambling or back-exchange is minimal. This new method for top-down HDX-MS enables real-time kinetic data measurements under physiological conditions, similar to those obtained using NMR, with comparable spatial resolution and significantly better sensitivity.
Figures



Similar articles
-
Determination of Backbone Amide Hydrogen Exchange Rates of Cytochrome c Using Partially Scrambled Electron Transfer Dissociation Data.J Am Soc Mass Spectrom. 2018 May;29(5):989-1001. doi: 10.1007/s13361-018-1892-3. Epub 2018 Mar 2. J Am Soc Mass Spectrom. 2018. PMID: 29500740
-
Real-time HD Exchange Kinetics of Proteins from Buffered Aqueous Solution with Electrothermal Supercharging and Top-Down Tandem Mass Spectrometry.J Am Soc Mass Spectrom. 2016 Jun;27(6):1019-27. doi: 10.1007/s13361-016-1350-z. Epub 2016 Feb 26. J Am Soc Mass Spectrom. 2016. PMID: 26919868 Free PMC article.
-
Nonuniform isotope patterns produced by collision-induced dissociation of homogeneously labeled ubiquitin: implications for spatially resolved hydrogen/deuterium exchange ESI-MS studies.Anal Chem. 2008 Jun 1;80(11):4078-86. doi: 10.1021/ac8001963. Epub 2008 May 7. Anal Chem. 2008. PMID: 18459737
-
Measuring the hydrogen/deuterium exchange of proteins at high spatial resolution by mass spectrometry: overcoming gas-phase hydrogen/deuterium scrambling.Acc Chem Res. 2014 Oct 21;47(10):3018-27. doi: 10.1021/ar500194w. Epub 2014 Aug 29. Acc Chem Res. 2014. PMID: 25171396 Review.
-
Mass spectrometric approaches using electrospray ionization charge states and hydrogen-deuterium exchange for determining protein structures and their conformational changes.Mol Cell Proteomics. 2004 Jan;3(1):10-23. doi: 10.1074/mcp.R300010-MCP200. Epub 2003 Nov 16. Mol Cell Proteomics. 2004. PMID: 14623985 Review.
Cited by
-
High-Resolution HDX-MS of Cytochrome c Using Pepsin/Fungal Protease Type XIII Mixed Bed Column.J Am Soc Mass Spectrom. 2019 Feb;30(2):227-234. doi: 10.1007/s13361-018-2087-7. Epub 2018 Oct 29. J Am Soc Mass Spectrom. 2019. PMID: 30374663
-
Construction of a Dual Protease Column, Subzero (-30 °C) Chromatography System and Multi-channel Precision Temperature Controller for Hydrogen-Deuterium Exchange Mass Spectrometry.J Res Natl Inst Stand Technol. 2020 Aug 12;vol:125025. doi: 10.6028/jres.125.025. eCollection 2020. J Res Natl Inst Stand Technol. 2020. PMID: 35573859 Free PMC article.
-
Anions in electrothermal supercharging of proteins with electrospray ionization follow a reverse Hofmeister series.Anal Chem. 2014 Feb 4;86(3):1640-7. doi: 10.1021/ac403398j. Epub 2014 Jan 24. Anal Chem. 2014. PMID: 24410546 Free PMC article.
-
Mass spectrometry-based methods to study protein architecture and dynamics.Protein Sci. 2013 May;22(5):530-44. doi: 10.1002/pro.2238. Epub 2013 Mar 26. Protein Sci. 2013. PMID: 23436701 Free PMC article. Review.
-
Charge enhancement of single-stranded DNA in negative electrospray ionization using the supercharging reagent meta-nitrobenzyl alcohol.J Am Soc Mass Spectrom. 2013 Dec;24(12):1988-96. doi: 10.1007/s13361-013-0732-8. Epub 2013 Sep 13. J Am Soc Mass Spectrom. 2013. PMID: 24030289
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

