Haploinsufficiency in peptidylglycine alpha-amidating monooxygenase leads to altered synaptic transmission in the amygdala and impaired emotional responses
- PMID: 20943906
- PMCID: PMC2975325
- DOI: 10.1523/JNEUROSCI.2200-10.2010
Haploinsufficiency in peptidylglycine alpha-amidating monooxygenase leads to altered synaptic transmission in the amygdala and impaired emotional responses
Abstract
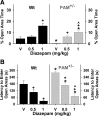
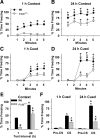
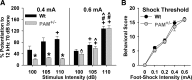
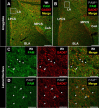
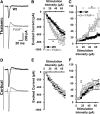
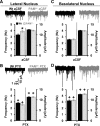
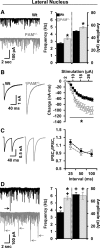
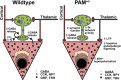
The mammalian amygdala expresses various neuropeptides whose signaling has been implicated in emotionality. Many neuropeptides require amidation for full activation by peptidylglycine α-amidating monooxygenase (PAM), a transmembrane vesicular cuproenzyme and regulator of the secretory pathway. Mice heterozygous for the Pam gene (PAM(+/-)) exhibit physiological and behavioral abnormalities related to specific peptidergic pathways. In the present study, we evaluated emotionality and examined molecular and cellular responses that characterize neurophysiological differences in the PAM(+/-) amygdala. PAM(+/-) mice presented with anxiety-like behaviors in the zero maze that were alleviated by diazepam. PAM(+/-) animals were deficient in short- and long-term contextual and cued fear conditioning and required higher shock intensities to establish fear-potentiated startle than their wild-type littermates. Immunohistochemical analysis of the amygdala revealed PAM expression in pyramidal neurons and local interneurons that synthesize GABA. We performed whole-cell recordings of pyramidal neurons in the PAM(+/-) amygdala to elucidate neurophysiological correlates of the fear behavioral phenotypes. Consistent with these observations, thalamic afferent synapses in the PAM(+/-) lateral nucleus were deficient in long-term potentiation. This deficit was apparent in the absence and presence of the GABA(A) receptor antagonist picrotoxin and was abolished when both GABA(A) and GABA(B) receptors were blocked. Both evoked and spontaneous excitatory signals were enhanced in the PAM(+/-) lateral nucleus. Phasic GABAergic signaling was also augmented in the PAM(+/-) amygdala, and this difference comprised activity-independent and -dependent components. These physiological findings represent perturbations in the PAM(+/-) amygdala that may underlie the aberrant emotional responses in the intact animal.
Figures


Similar articles
-
In vivo and in vitro analyses of amygdalar function reveal a role for copper.J Neurophysiol. 2014 May;111(10):1927-39. doi: 10.1152/jn.00631.2013. Epub 2014 Feb 19. J Neurophysiol. 2014. PMID: 24554785 Free PMC article.
-
Peptidylglycine α-amidating monooxygenase heterozygosity alters brain copper handling with region specificity.J Neurochem. 2013 Dec;127(5):605-19. doi: 10.1111/jnc.12438. Epub 2013 Oct 13. J Neurochem. 2013. PMID: 24032518 Free PMC article.
-
Pam heterozygous mice reveal essential role for Cu in amygdalar behavioral and synaptic function.Ann N Y Acad Sci. 2014 May;1314(1):15-23. doi: 10.1111/nyas.12378. Epub 2014 Mar 4. Ann N Y Acad Sci. 2014. PMID: 24593825 Free PMC article.
-
60 YEARS OF POMC: From POMC and α-MSH to PAM, molecular oxygen, copper, and vitamin C.J Mol Endocrinol. 2016 May;56(4):T63-76. doi: 10.1530/JME-15-0266. Epub 2015 Dec 14. J Mol Endocrinol. 2016. PMID: 26667899 Free PMC article. Review.
-
Peptidylglycine alpha-amidating monooxygenase: a multifunctional protein with catalytic, processing, and routing domains.Protein Sci. 1993 Apr;2(4):489-97. doi: 10.1002/pro.5560020401. Protein Sci. 1993. PMID: 8518727 Free PMC article. Review.
Cited by
-
Germline loss-of-function PAM variants are enriched in subjects with pituitary hypersecretion.medRxiv [Preprint]. 2023 Jan 20:2023.01.20.23284646. doi: 10.1101/2023.01.20.23284646. medRxiv. 2023. Update in: Front Endocrinol (Lausanne). 2023 Jun 14;14:1166076. doi: 10.3389/fendo.2023.1166076. PMID: 36711613 Free PMC article. Updated. Preprint.
-
Germline loss-of-function PAM variants are enriched in subjects with pituitary hypersecretion.Front Endocrinol (Lausanne). 2023 Jun 14;14:1166076. doi: 10.3389/fendo.2023.1166076. eCollection 2023. Front Endocrinol (Lausanne). 2023. PMID: 37388215 Free PMC article.
-
Stress-Related Neuronal Clusters in Sublenticular Extended Amygdala of Basal Forebrain Show Individual Differences of Positions.Front Neural Circuits. 2020 May 28;14:29. doi: 10.3389/fncir.2020.00029. eCollection 2020. Front Neural Circuits. 2020. PMID: 32547372 Free PMC article.
-
Signaling from the secretory granule to the nucleus.Crit Rev Biochem Mol Biol. 2012 Jul-Aug;47(4):391-406. doi: 10.3109/10409238.2012.694845. Epub 2012 Jun 8. Crit Rev Biochem Mol Biol. 2012. PMID: 22681236 Free PMC article. Review.
-
In vivo and in vitro analyses of amygdalar function reveal a role for copper.J Neurophysiol. 2014 May;111(10):1927-39. doi: 10.1152/jn.00631.2013. Epub 2014 Feb 19. J Neurophysiol. 2014. PMID: 24554785 Free PMC article.
References
-
- Baldwin GS, Patel O, Shulkes A. Phylogenetic analysis of the sequences of gastrin-releasing peptide and its receptors: biological implications. Regul Pept. 2007;143:1–14. - PubMed
-
- Bale TL, Vale WW. CRF and CRF receptors: role in stress responsivity and other behaviors. Annu Rev Pharmacol Toxicol. 2004;44:525–557. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous
