A homolog of the vertebrate pituitary adenylate cyclase-activating polypeptide is both necessary and instructive for the rapid formation of associative memory in an invertebrate
- PMID: 20943917
- PMCID: PMC6633720
- DOI: 10.1523/JNEUROSCI.2577-10.2010
A homolog of the vertebrate pituitary adenylate cyclase-activating polypeptide is both necessary and instructive for the rapid formation of associative memory in an invertebrate
Abstract
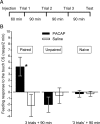
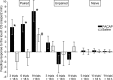
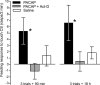
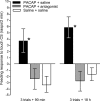
Similar to other invertebrate and vertebrate animals, cAMP-dependent signaling cascades are key components of long-term memory (LTM) formation in the snail Lymnaea stagnalis, an established experimental model for studying evolutionarily conserved molecular mechanisms of long-term associative memory. Although a great deal is already known about the signaling cascades activated by cAMP, the molecules involved in the learning-induced activation of adenylate cyclase (AC) in Lymnaea remained unknown. Using matrix-assisted laser desorption/ionization time-of-flight mass spectroscopy in combination with biochemical and immunohistochemical methods, recently we have obtained evidence for the existence of a Lymnaea homolog of the vertebrate pituitary adenylate cyclase-activating polypeptide (PACAP) and for the AC-activating effect of PACAP in the Lymnaea nervous system. Here we first tested the hypothesis that PACAP plays an important role in the formation of robust LTM after single-trial classical food-reward conditioning. Application of the PACAP receptor antagonist PACAP6-38 around the time of single-trial training with amyl acetate and sucrose blocked associative LTM, suggesting that in this "strong" food-reward conditioning paradigm the activation of AC by PACAP was necessary for LTM to form. We found that in a "weak" multitrial food-reward conditioning paradigm, lip touch paired with sucrose, memory formation was also dependent on PACAP. Significantly, systemic application of PACAP at the beginning of multitrial tactile conditioning accelerated the formation of transcription-dependent memory. Our findings provide the first evidence to show that in the same nervous system PACAP is both necessary and instructive for fast and robust memory formation after reward classical conditioning.
Figures

Similar articles
-
Reversal of age-related learning deficiency by the vertebrate PACAP and IGF-1 in a novel invertebrate model of aging: the pond snail (Lymnaea stagnalis).J Gerontol A Biol Sci Med Sci. 2014 Nov;69(11):1331-8. doi: 10.1093/gerona/glu068. Epub 2014 May 20. J Gerontol A Biol Sci Med Sci. 2014. PMID: 24846768 Free PMC article.
-
Pituitary adenylate cyclase activating polypeptide (PACAP) and its receptors are present and biochemically active in the central nervous system of the pond snail Lymnaea stagnalis.J Mol Neurosci. 2010 Nov;42(3):464-71. doi: 10.1007/s12031-010-9361-x. Epub 2010 Apr 16. J Mol Neurosci. 2010. PMID: 20396976
-
Same same, but different: exploring the enigmatic role of the pituitary adenylate cyclase-activating polypeptide (PACAP) in invertebrate physiology.J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2024 Nov;210(6):909-925. doi: 10.1007/s00359-024-01706-5. Epub 2024 Jun 28. J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2024. PMID: 38940930 Free PMC article.
-
[Physiological significance of pituitary adenylate cyclase-activating polypeptide (PACAP) in the nervous system].Yakugaku Zasshi. 2002 Dec;122(12):1109-21. doi: 10.1248/yakushi.122.1109. Yakugaku Zasshi. 2002. PMID: 12510388 Review. Japanese.
-
Pituitary adenylate cyclase-activating polypeptide (PACAP) in the hypothalamic-pituitary-gonadal axis: a review of the literature.Reprod Sci. 2013 Aug;20(8):857-71. doi: 10.1177/1933719112466310. Epub 2012 Dec 10. Reprod Sci. 2013. PMID: 23232965 Review.
Cited by
-
Function of insulin in snail brain in associative learning.J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2015 Oct;201(10):969-81. doi: 10.1007/s00359-015-1032-5. Epub 2015 Aug 2. J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2015. PMID: 26233474 Review.
-
Reversal of age-related learning deficiency by the vertebrate PACAP and IGF-1 in a novel invertebrate model of aging: the pond snail (Lymnaea stagnalis).J Gerontol A Biol Sci Med Sci. 2014 Nov;69(11):1331-8. doi: 10.1093/gerona/glu068. Epub 2014 May 20. J Gerontol A Biol Sci Med Sci. 2014. PMID: 24846768 Free PMC article.
-
A flavonoid, quercetin, is capable of enhancing long-term memory formation if encountered at different times in the learning, memory formation, and memory recall continuum.J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2022 Mar;208(2):253-265. doi: 10.1007/s00359-021-01522-1. Epub 2021 Nov 24. J Comp Physiol A Neuroethol Sens Neural Behav Physiol. 2022. PMID: 34820709
-
The unlimited potential of the great pond snail, Lymnaea stagnalis.Elife. 2020 Jun 16;9:e56962. doi: 10.7554/eLife.56962. Elife. 2020. PMID: 32539932 Free PMC article.
-
Interneuronal mechanisms for learning-induced switch in a sensory response that anticipates changes in behavioral outcomes.Curr Biol. 2021 Apr 26;31(8):1754-1761.e3. doi: 10.1016/j.cub.2021.01.072. Epub 2021 Feb 10. Curr Biol. 2021. PMID: 33571436 Free PMC article.
References
-
- Abrams TW, Castellucci VF, Camardo JS, Kandel ER, Lloyd PE. Two endogenous neuropeptides modulate the gill and siphon withdrawal reflex in Aplysia by presynaptic facilitation involving cAMP-dependent closure of a serotonin-sensitive potassium channel. Proc Natl Acad Sci U S A. 1984;81:7956–7960. - PMC - PubMed
-
- Alexander J, Jr, Audesirk TE, Audesirk GJ. One-trial reward learning in the snail Lymnaea stagnalis. J Neurobiol. 1984;15:67–72. - PubMed
-
- Bacskai BJ, Hochner B, Mahaut-Smith M, Adams SR, Kaang BK, Kandel ER, Tsien RY. Spatially resolved dynamics of cAMP and protein kinase A subunits in Aplysia sensory neurons. Science. 1993;260:222–266. - PubMed
-
- Bhattacharya A, Lakhman SS, Singh S. Modulation of L-type calcium channels in Drosophila via a pituitary adenylyl cyclase-activating polypeptide (PACAP)-mediated pathway. J Biol Chem. 2004;279:37291–37297. - PubMed
-
- Brembs B, Lorenzetti FD, Reyes FD, Baxter DA, Byrne JH. Operant reward learning in Aplysia: neuronal correlates and mechanisms. Science. 2002;296:1706–1709. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
