Cell death or survival promoted by alternative isoforms of ErbB4
- PMID: 20943952
- PMCID: PMC2993754
- DOI: 10.1091/mbc.E10-04-0332
Cell death or survival promoted by alternative isoforms of ErbB4
Abstract
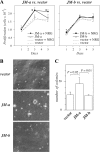
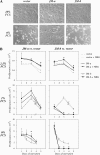
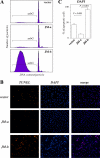
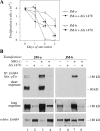
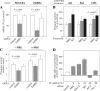
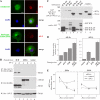
The significance of ErbB4 in tumor biology is poorly understood. The ERBB4 gene is alternatively spliced producing juxtamembrane (JM-a and JM-b) and cytoplasmic (CYT-1 and CYT-2) isoforms. Here, signaling via the two alternative ErbB4 JM isoforms (JM-a CYT-2 and JM-b CYT-2) was compared. Fibroblasts expressing ErbB4 JM-a demonstrated enhanced ErbB4 autophosphorylation, growth, and survival. In contrast, cells overexpressing ErbB4 JM-b underwent starvation-induced death. Both pro- and antisurvival responses to the two ErbB4 isoforms were sensitive to an ErbB kinase inhibitor. Platelet-derived growth factor receptor-alpha (PDGFRA) was identified as an ErbB4 target gene that was differentially regulated by the two ErbB4 isoforms. The soluble intracellular domain of ErbB4, released from the JM-a but not from the JM-b isoform, associated with the transcription factor AP-2 and promoted its potential to enhance PDGFRA transcription. Survival of cells expressing JM-a was suppressed by targeting either PDGFR-α or AP-2, whereas cells expressing JM-b were rescued from cell death by the PDGFR-α agonist, PDGF-BB. These findings indicate that two alternative ErbB4 isoforms may promote antagonistic cellular responses and suggest that pharmacological inhibition of ErbB4 kinase activity may lead to either suppression or promotion of cellular growth.
Figures


Similar articles
-
Overexpression of ERBB4 JM-a CYT-1 and CYT-2 isoforms in transgenic mice reveals isoform-specific roles in mammary gland development and carcinogenesis.Breast Cancer Res. 2014 Dec 17;16(6):501. doi: 10.1186/s13058-014-0501-z. Breast Cancer Res. 2014. PMID: 25516216 Free PMC article.
-
Proteolytic cleavage and phosphorylation of a tumor-associated ErbB4 isoform promote ligand-independent survival and cancer cell growth.Mol Biol Cell. 2006 Jan;17(1):67-79. doi: 10.1091/mbc.e05-05-0402. Epub 2005 Oct 26. Mol Biol Cell. 2006. PMID: 16251361 Free PMC article.
-
ErbB4 isoforms selectively regulate growth factor induced Madin-Darby canine kidney cell tubulogenesis.Mol Biol Cell. 2007 Nov;18(11):4446-56. doi: 10.1091/mbc.e07-03-0223. Epub 2007 Aug 29. Mol Biol Cell. 2007. PMID: 17761534 Free PMC article.
-
Erbb4 and its isoforms: selective regulation of growth factor responses by naturally occurring receptor variants.Trends Cardiovasc Med. 2000 Oct;10(7):304-10. doi: 10.1016/s1050-1738(01)00065-2. Trends Cardiovasc Med. 2000. PMID: 11343971 Review.
-
Function of ERBB4 is determined by alternative splicing.Cell Cycle. 2011 Aug 15;10(16):2647-57. doi: 10.4161/cc.10.16.17194. Epub 2011 Aug 15. Cell Cycle. 2011. PMID: 21811097 Review.
Cited by
-
Activating ERBB4 mutations in non-small cell lung cancer.Oncogene. 2016 Mar 10;35(10):1283-91. doi: 10.1038/onc.2015.185. Epub 2015 Jun 8. Oncogene. 2016. PMID: 26050618
-
Target HER four in breast cancer?Oncotarget. 2019 May 7;10(34):3147-3150. doi: 10.18632/oncotarget.26867. eCollection 2019 May 7. Oncotarget. 2019. PMID: 31139328 Free PMC article.
-
Nuclear HER4 mediates acquired resistance to trastuzumab and is associated with poor outcome in HER2 positive breast cancer.Oncotarget. 2014 Aug 15;5(15):5934-49. doi: 10.18632/oncotarget.1904. Oncotarget. 2014. PMID: 25153719 Free PMC article.
-
ERBB4 Expression in Ovarian Serous Carcinoma Resistant to Platinum-Based Therapy.Cancer Control. 2017 Jan;24(1):89-95. doi: 10.1177/107327481702400115. Cancer Control. 2017. PMID: 28178720 Free PMC article.
-
The Yin and Yang of ERBB4: Tumor Suppressor and Oncoprotein.Pharmacol Rev. 2022 Jan;74(1):18-47. doi: 10.1124/pharmrev.121.000381. Pharmacol Rev. 2022. PMID: 34987087 Free PMC article. Review.
References
-
- Afink G. B., Nister M., Stassen B. H., Joosten P. H., Rademakers P. J., Bongcam-Rudloff E., Van Zoelen E. J., Mosselman S. Molecular cloning and functional characterization of the human platelet-derived growth factor alpha receptor gene promoter. Oncogene. 1995;8:1667–1672. - PubMed
-
- Alaoui-Jamali M. A., Song D. J., Benlimame N., Yen L., Deng X., Hernandez-Perez M., Wang T. Regulation of multiple tumor microenvironment markers by overexpression of single or paired combinations of ErbB receptors. Cancer Res. 2003;13:3764–3774. - PubMed
-
- Aqeilan R. I., Donati V., Palamarchuk A., Trapasso F., Kaou M., Pekarsky Y., Sudol M., Croce C. M. WW domain-containing proteins, WWOX and YAP, compete for interaction with ErbB-4 and modulate its transcriptional function. Cancer Res. 2005;15:6764–6772. - PubMed
-
- Aqeilan R. I., Palamarchuk A., Weigel R. J., Herrero J. J., Pekarsky Y., Croce C. M. Physical and functional interactions between the Wwox tumor suppressor protein and the AP-2gamma transcription factor. Cancer Res. 2004a;22:8256–8261. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

