Interactions between the immune and nervous systems in pain
- PMID: 20948535
- PMCID: PMC3077564
- DOI: 10.1038/nm.2234
Interactions between the immune and nervous systems in pain
Abstract
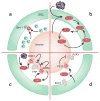
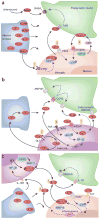
Immune cells and glia interact with neurons to alter pain sensitivity and to mediate the transition from acute to chronic pain. In response to injury, resident immune cells are activated and blood-borne immune cells are recruited to the site of injury. Immune cells not only contribute to immune protection but also initiate the sensitization of peripheral nociceptors. Through the synthesis and release of inflammatory mediators and interactions with neurotransmitters and their receptors, the immune cells, glia and neurons form an integrated network that coordinates immune responses and modulates the excitability of pain pathways. The immune system also reduces sensitization by producing immune-derived analgesic and anti-inflammatory or proresolution agents. A greater understanding of the role of the immune system in pain processing and modulation reveals potential targets for analgesic drug development and new therapeutic opportunities for managing chronic pain.
Conflict of interest statement
The authors declare no competing financial interests.
Figures

References
-
- DeLeo JA, Tanga FY, Tawfik VL. Neuroimmune activation and neuroinflammation in chronic pain and opioid tolerance/hyperalgesia. Neuroscientist. 2004;10:40–52. - PubMed
-
- Watkins LR, Maier SF. Immune regulation of central nervous system functions: from sickness responses to pathological pain. J Intern Med. 2005;257:139–155. - PubMed
-
- Scholz J, Woolf CJ. The neuropathic pain triad: neurons, immune cells and glia. Nat Neurosci. 2007;10:1361–1368. - PubMed
-
- Thacker MA, Clark AK, Marchand F, McMahon SB. Pathophysiology of peripheral neuropathic pain: immune cells and molecules. Anesth Analg. 2007;105:838–847. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

