Effect of feeding status on adjuvant arthritis severity, cachexia, and insulin sensitivity in male Lewis rats
- PMID: 20953376
- PMCID: PMC2952917
- DOI: 10.1155/2010/398026
Effect of feeding status on adjuvant arthritis severity, cachexia, and insulin sensitivity in male Lewis rats
Abstract
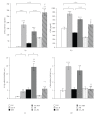
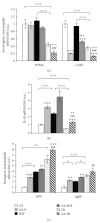
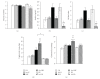
We studied the effect of food restriction, overfeeding, and normofeeding on cachexia, inflammatory and metabolic parameters, and insulin sensitivity in chronic adjuvant arthritis (AA) in rats. Food restriction during AA increased circulating ghrelin, corticosterone, decreased leptin, and ameliorated arthrogram score and systemic inflammation compared to normofeeding. Overfeeding worsened arthrogram score and systemic inflammation, and led to lipid accumulation in the liver, but not to alterations of adipokine and ghrelin plasma levels relative to normofeeding. Independently of feeding status, AA induced cachexia, in which modulation of mRNA expressions for appetite-regulating neuropeptides (NPY, AgRP, POMC, CART) in the arcuate nucleus (ARC) does not play a primary role. The overexpression of IL-1β mRNA in the ARC suggests its role in the mechanisms of impaired energy balance during AA under all feeding conditions. Normal HOMA index in all arthritic groups does not indicate the development of insulin resistance by feeding interventions in these rats.
Figures


References
-
- Bays HE, González-Campoy JM, Bray GA, et al. Pathogenic potential of adipose tissue and metabolic consequences of adipocyte hypertrophy and increased visceral adiposity. Expert Review of Cardiovascular Therapy. 2008;6(3):343–368. - PubMed
-
- Bartfai T, Waalen J, Buxbaum JN. Adipose tissue as a modulator of clinical inflammation: does obesity reduce the prevalence of rheumatoid arthritis? Journal of Rheumatology. 2007;34(3):488–492. - PubMed
-
- Summers GD, Deighton CM, Rennie MJ, Booth AH. Rheumatoid cachexia: a clinical perspective. Rheumatology. 2008;47(8):1124–1131. - PubMed
-
- Van Der Helm-van Mil AHM, Van Der Kooij SM, Allaart CF, Toes REM, Huizinga TWJ. A high body mass index has a protective effect on the amount of joint destruction in small joints in early rheumatoid arthritis. Annals of the Rheumatic Diseases. 2008;67(6):769–774. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

