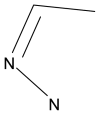
3D-QSAR and molecular docking studies on fused pyrazoles as p38α mitogen-activated protein kinase inhibitors
- PMID: 20957100
- PMCID: PMC2956100
- DOI: 10.3390/ijms11093357
3D-QSAR and molecular docking studies on fused pyrazoles as p38α mitogen-activated protein kinase inhibitors
Abstract
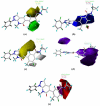
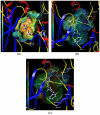
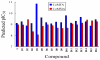
The p38α mitogen-activated protein kinase (MAPK) has become an attractive target for the treatment of many diseases such as rheumatoid arthritis, inflammatory bowel disease and Crohn's disease. In this paper, 3D-QSAR and molecular docking studies were performed on 59 p38α MAPK inhibitors. Comparative molecular field analysis (CoMFA) and comparative molecular similarity indices analysis (CoMSIA) were applied to determine the structural requirements for potency in inhibiting p38α MAPK. The resulting model of CoMFA and CoMSIA exhibited good r(2) (cv) values of 0.725 and 0.609, and r(2) values of 0.961 and 0.905, respectively. Molecular docking was used to explore the binding mode between the inhibitors and p38α MAPK. We have accordingly designed a series of novel p38α MAPK inhibitors by utilizing the structure-activity relationship (SAR) results revealed in the present study, which were predicted with excellent potencies in the developed models. The results provided a useful guide to design new compounds for p38α MAPK inhibitors.
Keywords: 3D-QSAR; CoMFA; CoMSIA; docking; p38α mitogen-activated protein kinase.
Figures



Similar articles
-
Molecular modeling of p38α mitogen-activated protein kinase inhibitors through 3D-QSAR and molecular dynamics simulations.J Chem Inf Model. 2013 Jul 22;53(7):1775-86. doi: 10.1021/ci4000085. Epub 2013 Jul 10. J Chem Inf Model. 2013. PMID: 23808966
-
3D-QSAR and molecular docking analysis of biphenyl amide derivatives as p38α mitogen-activated protein kinase inhibitors.Mol Divers. 2012 May;16(2):377-88. doi: 10.1007/s11030-011-9353-y. Epub 2012 Jan 7. Mol Divers. 2012. PMID: 22228035
-
Structure-based 3D-QSAR models and dynamics analysis of novel N-benzyl pyridinone as p38α MAP kinase inhibitors for anticytokine activity.J Mol Graph Model. 2012 Jun;36:48-61. doi: 10.1016/j.jmgm.2012.02.003. Epub 2012 Mar 6. J Mol Graph Model. 2012. PMID: 22534481
-
A Comprehensive Structural Overview of p38α MAPK in Complex with Type I Inhibitors.ChemMedChem. 2015 Jun;10(6):957-69. doi: 10.1002/cmdc.201500030. Epub 2015 Apr 9. ChemMedChem. 2015. PMID: 26012502 Review.
-
CNS-Active p38α MAPK Inhibitors for the Management of Neuroinflammatory Diseases: Medicinal Chemical Properties and Therapeutic Capabilities.Mol Neurobiol. 2024 Jul;61(7):3911-3933. doi: 10.1007/s12035-023-03829-3. Epub 2023 Dec 2. Mol Neurobiol. 2024. PMID: 38041716 Review.
Cited by
-
Insight into the interactions between novel isoquinolin-1,3-dione derivatives and cyclin-dependent kinase 4 combining QSAR and molecular docking.PLoS One. 2014 Apr 10;9(4):e93704. doi: 10.1371/journal.pone.0093704. eCollection 2014. PLoS One. 2014. PMID: 24722522 Free PMC article.
-
3D-QSAR studies and molecular docking on [5-(4-amino-1H-benzoimidazol-2-yl)-furan-2-yl]-phosphonic acid derivatives as fructose-1,6-biphophatase inhibitors.J Comput Aided Mol Des. 2010 Dec;24(12):993-1008. doi: 10.1007/s10822-010-9391-z. Epub 2010 Oct 20. J Comput Aided Mol Des. 2010. PMID: 20960032
-
Integrating sampling techniques and inverse virtual screening: toward the discovery of artificial peptide-based receptors for ligands.Mol Divers. 2016 May;20(2):421-38. doi: 10.1007/s11030-015-9648-5. Epub 2015 Nov 9. Mol Divers. 2016. PMID: 26553204
-
Understanding the structure-activity relationship of betulinic acid derivatives as anti-HIV-1 agents by using 3D-QSAR and docking.J Mol Model. 2011 Jul;17(7):1643-59. doi: 10.1007/s00894-010-0870-x. Epub 2010 Oct 27. J Mol Model. 2011. PMID: 20978914
-
Molecular modeling studies on phosphonic acid-containing thiazole derivatives: design for fructose-1,6-bisphosphatase inhibitors.J Mol Model. 2012 Mar;18(3):973-90. doi: 10.1007/s00894-011-1134-0. Epub 2011 Jun 5. J Mol Model. 2012. PMID: 21644014
References
-
- Pettus LH, Wurz RP, Xu S, Herberich B, Henkle B, Liu Q, McBride HJ, Mu S, Plant MH, Saris CJM, Sherman L, Wong LM, Chmait S, Lee MR, Mohr C, Hsieh F, Tasker AS. Discovery and evaluation of 7-alkyl-1,5-bis-aryl-pyrazolopyridinones as highly potent, selective, and orally efficacious inhibitors of p38α mitogen-activated protein kinase. J. Med. Chem. 2010;53:2973–2985. - PubMed
-
- Wurz RP, Pettus LH, Henkle B, Sherman L, Plant M, Miner K, McBride HJ, Wong LM, Saris CJM, Lee MR, Chmait S, Mohr C, Hsieh F, Tasker AS. Part2: Structure-activity relationship (SAR) investigations of fused pyrazoles as potent, selective and orally available inhibitors of p38α mitogen-activated protein kinase. Bioorg. Med. Chem. Lett. 2010;20:1680–1684. - PubMed
-
- Wurz RP, Pettus LH, Xu S, Henkle B, Sherman L, Plant M, Miner K, McBride H, Wong LM, Saris CJM, Lee MR, Chmait S, Mohr C, Hsieh F, Tasker AS. Part 1: Structure-activity relationship (SAR) investigations of fused pyrazoles as potent, selective and orally available inhibitors of p38α mitogen-activated protein kinase. Bioorg. Med. Chem. Lett. 2009;19:4724–4728. - PubMed
-
- Hynes J, Jr, Dyckman AJ, Lin S, Wrobleski ST, Wu H, Gillooly KM, Kanner SB, Lonial H, Loo D, McIntyre KW, Pitt S, Shen DR, Shuster DJ, Yang S, Zhang R, Behnia K, Zhang H, Marathe PH, Doweyko AM, Tokarski JS, Sack JS, Pokross M, Kiefer SE, Newitt JA, Barrish JC, Dodd J, Schieven GL, Leftheris K. Design, synthesis, and anti-inflammatory properties of orally active 4-(phenylamino)-pyrrolo[2,1-f][1,2,4]triazine p38α mitogen-activated protein kinase inhibitors. J. Med. Chem. 2008;51:4–16. - PubMed
-
- Pettus LH, Xu S, Cao G-Q, Chakrabarti PP, Rzasa RM, Sham K, Wurz RP, Zhang D, Middleton S, Henkle B, Plant MH, Saris CJM, Sherman L, Wong LM, Powers DA, Tudor Y, Yu V, Lee MR, Syed R, Hsieh F, Tasker AS. 3-Amino-7-phthalazinylbenzoisoxazoles as a novel class of potent, selective, and orally available inhibitors of p38α mitogen-activated protein kinase. J. Med. Chem. 2008;51:6280–6292. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

